Access a toolkit of functional, consistent in vitro models to study neurodegenerative disease and neuroinflammation
Powered by opti-ox
Powered by opti-ox
Microglia, the resident macrophages of the CNS, play critical roles in neural function by regulating neurogenesis, synaptic remodelling, and serving as first responders to infection. Dysregulated microglia activity is highly implicated in the pathology of neurodegenerative diseases, including Alzheimer’s, Parkinson's, and Huntington's disease.
Studying microglia dysfunction is challenging, as many available models fail to recapitulate human-specific disease phenotypes. Primary human microglia are hard to isolate and vary between donors, immortalised cell lines such as HMC3 are prone to dedifferentiation and genetic drift, and traditional protocols for directed differentiation of hiPSCs are laborious resulting in the production of heterogeneous cultures with high batch-to-batch variability.
ioMicroglia provides a rapidly maturing, consistent, and functional source of human iPSC-derived microglia, bypassing these limitations. ioMicroglia are ready for experiments within 4 days of thawing and can be easily integrated into multi-cell culture models.
ioMicroglia rapidly acquire a homogeneous microglia phenotype upon thawing, as captured in this 10-day time course. This consistent and rapid maturation provides scientists with confidence in the reproducible generation of functional cultures in just a few days.
The HMC3 immortalised cell line has long been a widely used microglial model due to its ease of culture and scalability. However, it has some limitations that compromise its utility for translational research. HMC3 cells lack key microglial identity markers like P2RY12 and TREM2, possess a transcriptomic profile significantly different from primary microglia, and functionally demonstrate very weak or no phagocytic activity and a blunted cytokine response. In contrast, ioMicroglia provide a consistent, phenotypically accurate model, expressing key canonical markers and demonstrating robust phagocytic function and cytokine release profiles. Incorporating this human-relevant model offers a powerful opportunity to de-risk the drug discovery pipeline, filter out false positives, and increase the translational potential of preclinical findings.
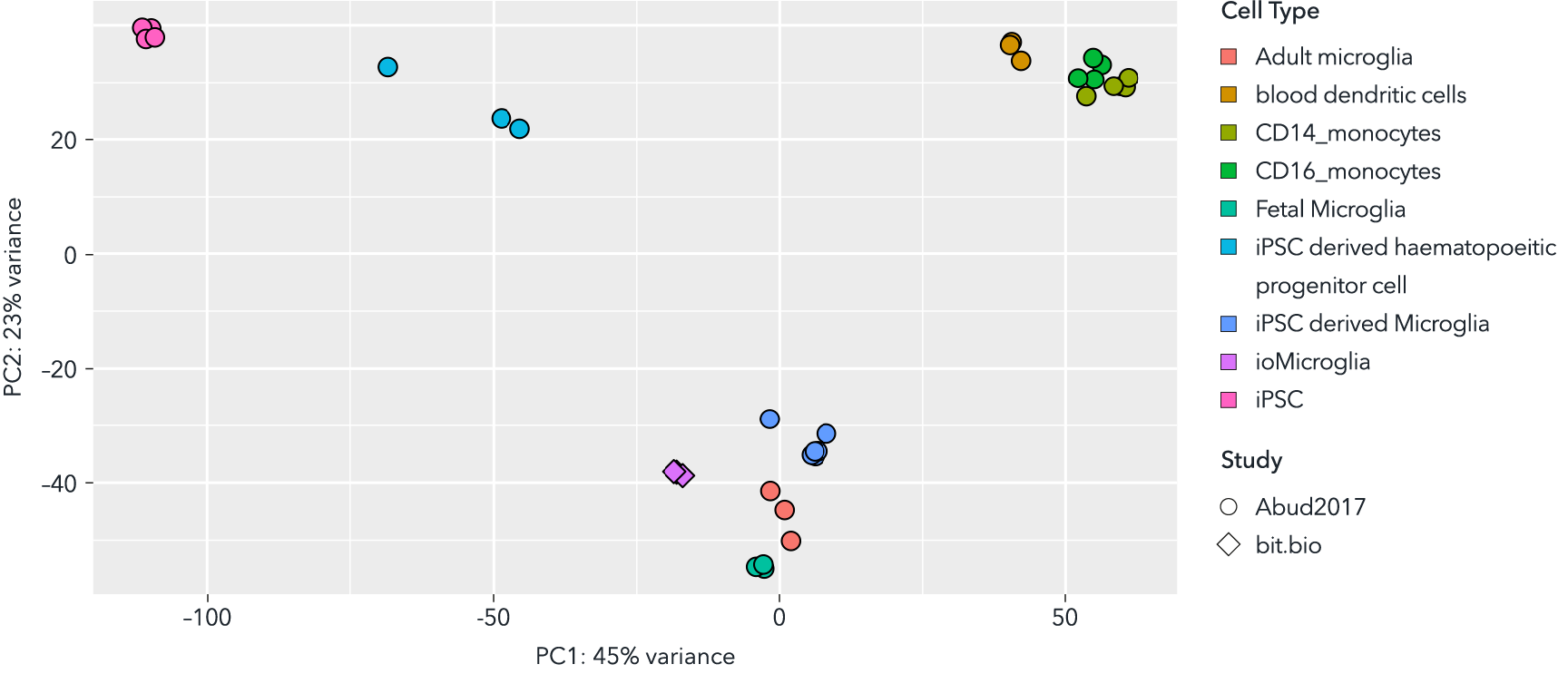
ioMicroglia closely mimic the molecular identity of human foetal and adult microglia. They overcome the key limitations of sourcing constraints and donor variability of primary cells as well as the heterogeneity and batch-to-batch inconsistency caused by complex differentiation protocols.
This principal component analysis (PCA) plot of bulk RNA sequencing data validates the transcriptomic identity of ioMicroglia. By integrating male donor-derived ioMicroglia with external data sets from Abud et al1., the analysis reveals that ioMicroglia exhibit a transcriptomic clustering with primary foetal and adult microglia. This demonstrates that ioMicroglia possess a molecular profile highly similar with in vivo human cells, providing an accessible, physiologically relevant model.

The cytokine release assay is a valuable tool for ensuring that microglia behave in vitro as they would in vivo. ioMicroglia secrete proinflammatory and anti-inflammatory cytokines (IL-6, TNFα, IL-1β,and IL-8) in response to stimuli such as Lipopolysaccharides (LPS) and Interferon Gamma (IFN-γ), and amyloidβ-42 (Aβ42).
ioMicroglia demonstrate robust and selective phagocytic activity while in a 10-day co-culture with ioGlutamatergic Neurons. As captured in this time-lapse, the microglia actively engulf pHrodo Red Zymosan particles without any adverse effects on neuronal morphology, giving scientists confidence in their ability to build stable, functionally validated co-culture models for studying specific glial-neuronal interactions.
bit.bio has optimised protocols for phagocytosis and cytokine release assays, enabling scientists to implement co-culture systems and perform these assays with ease.
.png?width=600&height=329&name=AB42%20Percentage%20positive-%20Male%20WT%20ApoE%20(1).png)
ioMicroglia have been engineered with early-onset Alzheimer’s disease mutations in APOE and TREM2 genes, providing a system to study disease-related phenotypes. These models demonstrate robust phagocytic activity against both E. coli and fluorescent Aβ42 particles. Furthermore, the cells exhibit a complex cytokine secretion profile (including IL-6, IL-8, and IL-1β) in response to inflammatory stimuli (LPS/IFN-γ) and Aβ42 oligomers. This consistent, human-relevant model allows scientists to study how risk genotypes modulate core microglial functions.

A quad-culture model integrates ioMicroglia, ioOligodendrocyte-like cells, ioGlutamatergic Neurons, and human iPSC-derived astrocytes, providing a physiological system to investigate the role of glial cells in neurodegenerative disease mechanisms.

Human brain organoids often lack a functional immune component, limiting their ability to capture neuroimmune interactions. This application note demonstrates how integrating ioMicroglia enables a more physiologically relevant human 3D model with measurable inflammatory signalling, cytokine release, and pharmacological modulation.

ioMicroglia have been engineered to constitutively express green fluorescent protein (GFP), offering scientists a fluorescent human microglia model ideal for co-culture with other neural cell types, enabling effortless tracking in multi-cellular systems.
Constitutive GFP expression throughout the cytosol facilitates live-cell imaging, enabling the assessment of cell motility and visualisation of microglial activation states. GFP-labelled human microglia streamline cell sorting workflows by eliminating the need for antibody staining.

CRISPRko-Ready ioMicroglia enable high-resolution pooled single-cell CRISPR knockout (scCRISPR) screens to systematically map gene function. In a targeted study focused on 110 neurodegeneration-linked genes, cells were transduced and subsequently challenged with LPS to induce a transcriptomic activation signature.
In this video, our scientist takes you through the step-by-step process of how to thaw, seed and culture ioMicroglia, which complements our expert scientist's top tips on understanding the dynamic morphology of microglia, handling cells gently, and using the right seeding density and media changes.

Matteo Zanella, PhD
Associate Research Leader | Charles River

Women are greatly underrepresented in drug development and clinical trials.
Introducing female-derived cells into the early stage of research and drug discovery can help to better address this disparity.
Key applications for Female ioMicroglia in neurodegeneration drug discovery
- Neuroinflammatory in vitro modelling
- Target ID and validation
- Compound screening
Discover the data

Access 20 neuronal disease models and 4 microglia disease models with a single co-culture protocol.
View the co-culture protocol
Explore ioGlutamatergic Neuron Disease Models
Explore ioMicroglia Disease Models
Human iPSC-derived microglia engineered to constitutively express GFP enable easy visualisation, tracking and isolation of cells in complex multi-cell cultures.
Discover the data


Built from our ioMicroglia Male and engineered to constitutively express Cas9.
With optimised guide RNA delivery protocols and high knockout efficiency, start measuring readouts from gene knockouts and CRISPR screens within days.
Save months of work by skipping complex cell line engineering and cell differentiation workflows.
Discover the data on CRISPRko-Ready ioMicroglia
bit.bio
Murgoci A.N. et al, 2026
BrainZell
Elise Malavasi, PhD
Principal Scientist
Concept Life Sciences
Raman, et al
bit.bio
2022
Smith, et al.
bit.bio
2024
Schmidt, et al
bit.bio
2024
Oosterveen et al.
bit.bio
2025
Bsibsi et al.
Courtesy of Charles River Laboratories
2024
Newman et al.
bit.bio
2024
Grabner et al.
bit.bio
2025
Veteleanu et al.
bit.bio
2025
Yates et al.
bit.bio
2025
Tatar Ozkan et al.
bit.bio
2025
Davenport A, Frolov T & Kotter M
Drug Discovery World
2020
Guerrisi S, et al.
BioRxiv
2026
Using ioGlutamatergic Neurons, ioGABAergic Neurons, ioMicroglia and ioMicroglia TREM2 R47H/R47H
Euan Yates | Scientist | bit.bio
Human Cell Forum 2025
Session 2 | bit.bio insider: Tools, tips, and what’s coming next
Dr Mariangela Iovino | Group Leader | Charles River
Dr Tony Oosterveen | Senior Scientist | bit.bio
Dr Matthias Pawlowski | Head, Dementia-Sensitive Hospital | University of Münster
Dr Malathi Raman | Senior Product Manager | bit.bio
Prof Roger Pedersen | Adjunct Professor and Senior Research Scientist at Stanford University
Dr Thomas Moreau | Director of Cell Biology Research | bit.bio
Antonella Santuccione Chadha, MD | Founder and CEO | Women’s Brain Foundation
Melanie Einsiedler, PhD | Scientific Contributor | Women’s Brain Foundation
Rebecca Northeast, PhD | Senior Product Manager | bit.bio
Microglia are the resident macrophages of the central nervous system (CNS), serving as the primary line of defence against infection and regulating critical homeostatic processes like neurogenesis and synaptic remodelling. The dysregulation of microglial activity plays a pivotal role in the pathogenesis of neurodegenerative disorders, specifically Alzheimer’s, Parkinson’s, and Huntington’s disease.
Immortalised microglial cell lines, such as HMC3, are prone to dedifferentiation and genetic drift, and often lack key microglial identity markers and show weak phagocytic activity. Human iPSC-derived microglia overcome these functional and phenotypic limitations by providing a physiologically relevant model that expresses canonical markers and avoids the instability associated with immortalised lines.
ioMicroglia recapitulate the transcriptomic profile of primary human foetal and adult microglia. Bulk RNA sequencing confirms that the gene expression profile of these cells cluster with primary datasets, validating their physiological relevance as a consistent alternative to donor-derived primary cells.
ioMicroglia, human iPSC-derived microglia, are compatible with standard functional assays, most notably phagocytosis and cytokine release screening. These cells demonstrate robust functional maturity by actively phagocytosing targets, such as E. coli and amyloid-beta (Aβ42) and secreting inflammatory cytokines (e.g., IL-6, TNFα) upon stimulation, enabling the precise modelling and quantification of neuroinflammatory states.
ioMicroglia, human iPSC-derived microglia, has been engineered with specific disease-related mutations, such as those found in the TREM2 and APOE genes associated with Alzheimer’s disease. By comparing these disease-relevant mutations to genetically matched wild-type controls, scientists can isolate how specific genotypes drive phenotypic changes, such as altered phagocytosis, in a controlled experimental setup.
Abud E, et al. "iPSC-Derived Human Microglia-like Cells to Study Neurological Diseases." Neuron, 2018; 94(2): 278-293, doi: 10.1016/j.neuron.2017.03.042
Woolf Z, et al. "In vitro models of microglia: a comparative study." Sci Rep 15, 2025; 15621, https://doi.org/10.1038/s41598-025-99867-z
Lloyd AF, et al. "Deep proteomic analysis of microglia reveals fundamental biological differences between model systems." Cell Reports, 2024. 43, issue 11, doi: 10.1016/j.celrep.2024.114908
Zhou Y., et al. "Human and mouse single-nucleus transcriptomics reveal TREM2-dependent and TREM2-independent cellular responses in Alzheimer's disease." Nat Med, 2020, 26(1):131-142, doi: 10.1038/s41591-019-0695-9