




























cat no | io1029
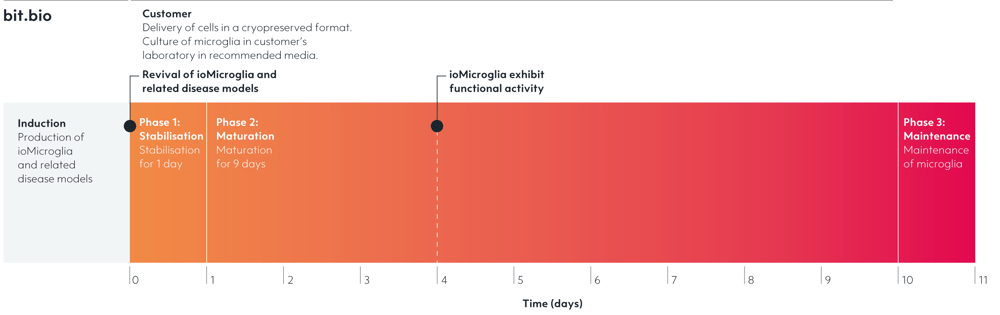
ioMicroglia Female
Female human iPSC donor-derived microglia
-
Cryopreserved human iPSC-derived cells powered by opti-ox, that are ready for experiments in 4 days
-
Ideal for in vitro multi-cellular neuroinflammation studies and modelling diversity in disease
-
Consistently perform key phagocytic and cytokine secretion functions, and are co-culture compatible

Human iPSC-derived microglia, female donor

ioMicroglia readily phagocytose by day 4 post-revival
Day 4, 6 and 10 ioMicroglia Female were incubated with 1 µg/0.33 cm2 pHrodo RED labelled E. coli particles for 24 hour. Images were acquired every 30 mins on the Incucyte looking at red fluorescence and phase contrast. The graphs display the proportion of cells phagocytosing (left), and the fluorescence intensity per cell displaying degree of phagocytosis (right). Three technical replicates were performed. Seeding density 60,000 cells/cm2.
Cells readily phagocytose by day 4 post-revival in a similar response as day 10, providing a experimental window to perform this key assay.

Phagocytosis of Amyloidβ-42 particles by female donor-derived ioMicroglia
Day 10 female donor-derived ioMicroglia were incubated with 500 nM AF488 labelled Amyloidβ-42 (AnaSpec) for 24 hours with images acquired every 30 mins on the Incucyte. The graphs display the proportion of cells phagocytosing (left), and the fluorescence intensity per cell displaying degree of phagocytosis (right). Three technical replicates were performed. Seeding density 60,000 cells/cm2.

Female donor-derived ioMicroglia demonstrate a measurable pro-inflammatory cytokine response by day 3
Day 3, 7, and 10 female donor-derived ioMicroglia were stimulated with LPS (100 ng/ml) and IFNɣ (20 ng/ml) for 24 hours, and supernatants and analysed using the MSD V-PLEX Proinflammatory Kit. Bars represent 3 technical replicates with standard error of mean.
A clear response is seen by day 3 post-revival, with the highest secretion levels observed at 10, demonstrating a wide time frame to perform this key assay.
View the cytokine release protocol used to generate this data.

Female donor-derived ioMicroglia show a higher pro-inflammatory cytokine response to Amyloidβ-42 stimulation compared to male donor-derived ioMicroglia
Day 10 male donor-derived ioMicroglia and female donor-derived ioMicroglia were stimulated for 24 hours with LPS (100 ng/ml) and IFNɣ (20 ng/ml), or synthetic Amyloidβ-42 oligomers (10 μM, StressMarq). Supernatants were harvested after 24 hours and analysed with using the MSD V-PLEX Proinflammatory Kit. Secretion levels were normalised to cell count per field of view (FOV) to account for variations in cell density. Seeding density 80,000 cells/cm2.
A clear response to Amyloidβ-42 is seen in both cell types, with background specific differences also observed.
View the cytokine release protocol used to generate this data.

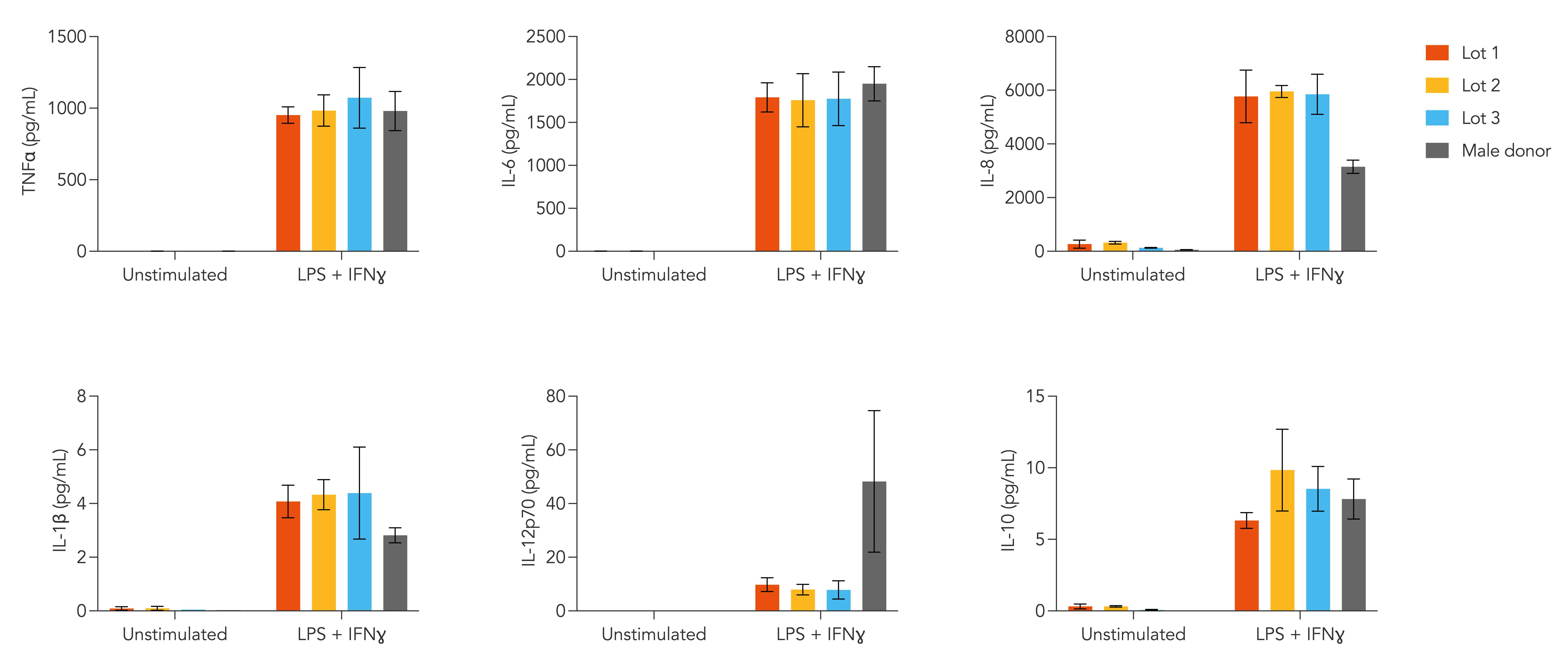
Female donor-derived ioMicroglia display a different pro-inflammatory cytokine response to male donor-derived cells
Day 10 female donor-derived ioMicroglia from three independent lots and male donor-derived ioMicroglia from one lot were stimulated with LPS (100 ng/ml) and IFNɣ (20 ng/ml) for 24 hours. Supernatants were harvested and analysed using MSD V-PLEX Proinflammatory Kit. Female donor-derived ioMicroglia secrete TNF⍺, IL-6, IL-8, IL-1b, IL-12p70 and IL-10 in response to stimuli, predominantly producing a pro-inflammatory response. This is consistent across three independent lots. Female donor-derived ioMicroglia show a higher level of secretion of IL-8 and IL-1β cytokines, and a lower level of IL-12p70 cytokine than male donor-derived ioMicroglia. Three technical replicates were performed per lot.
View the cytokine release protocol used to generate this data.

Female donor-derived ioMicroglia display a consistent degree of phagocytosis across lots
Day 10 female donor-derived ioMicroglia from three independent lots and male donor-derived ioMicroglia from one lot were incubated with pHrodo RED labelled Zymosan particles for 24 hours +/- cytochalasin D control. The graphs displays that the proportion of cells phagocytosing Zymosan particles (left), and the fluorescence intensity per cell displaying degree of phagocytosis (right) is consistent across three independent lots and that female donor-derived ioMicroglia cells display a higher proportion of phagocytosis than male donor-derived cells. Images were acquired every 30 mins on the Incucyte looking at red fluorescence and phase contrast. Three technical replicates were performed per lot.
Female donor-derived ioMicroglia phagocytose Zymosan particles
Representative video showing female donor-derived ioMicroglia (io1029) phagocytosing pHrodo Red labelled Zymosan particles. When female donor-derived ioMicroglia engulf these particles this causes the particles to fluoresce red, within the cells, due to the drop in pH in the phagolysosome. Live imaging was performed in 2-minute intervals over a time period of 2 hours using the 3D Cell Explorer 96focus Nanolive Imaging system.

Flow cytometry analysis reveals HMC3 cells do not express key microglia markers
Dot plots of cells expressing key Microglia associated markers P2RY12, CX3CR1, CD11b, CD14 and CD45. HMC3 cells show low expression of these markers. Male and female donor derived ioMicroglia demonstrate high expression, with minimal differences between the two backgrounds.

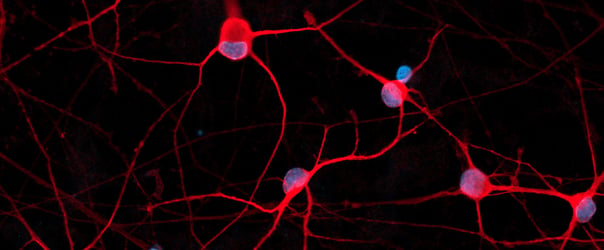
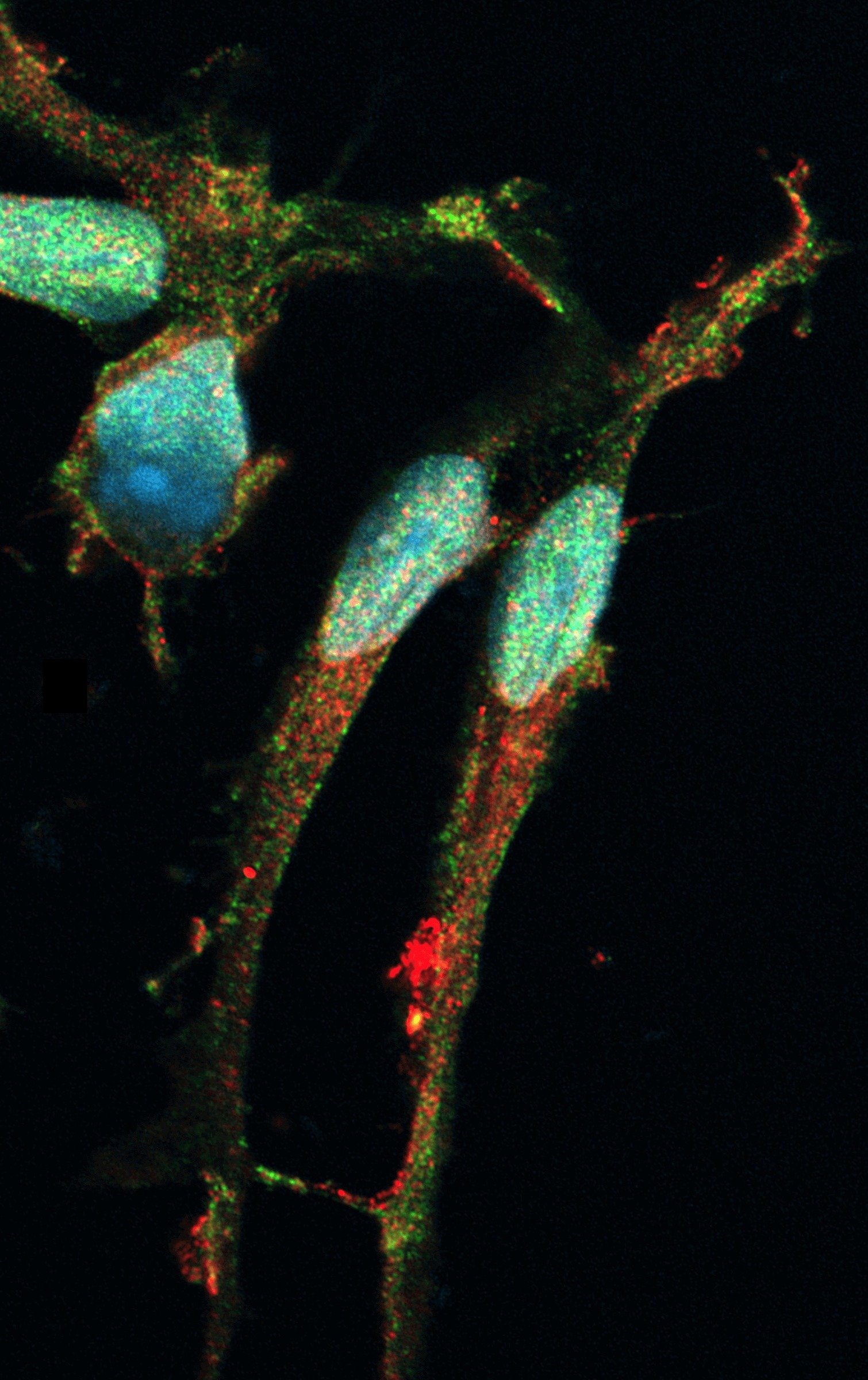
Female donor-derived ioMicroglia show key microglia marker expression
Immunofluorescent staining of day 10 female donor-derived ioMicroglia shows homogenous expression of IBA1 and TREM2, and a typical ramified morphology. DAPI counterstain (blue). Image taken at 10x magnification.

HMC3 cells show a blunted cytokine secretion response compared to ioMicroglia
Male and female donor-derived ioMicroglia demonstrate a much higher cytokine secretion response following immunostimulation. Cells were stimulated for 24 hours with LPS (100 ng/ml) and IFNɣ (20 ng/ml). Supernatants were harvested after 24 hours and analysed with using the MSD V-PLEX Proinflammatory Kit. Seeding density 60,000 cells/cm2. Bars represent 3 technical replicates with standard deviation of mean.

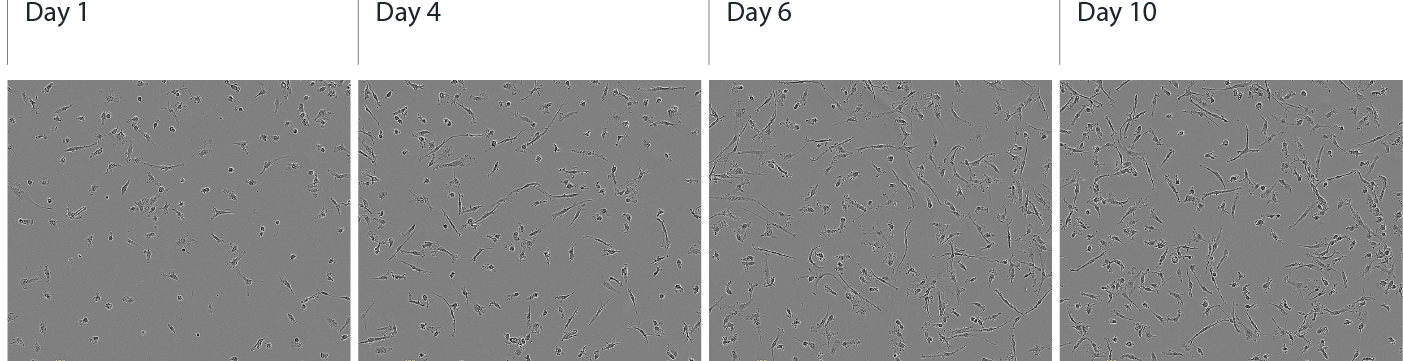
Female donor-derived ioMicroglia show ramified morphology by day 10
Rapid morphological changes in the female donor-derived cells upon reprogramming, with key ramified morphology first identified by day 4 and continuing through to day 10. Day 1 to 10 post-thawing; 100x magnification.
opti-ox precision deterministic programmed ioMicroglia from a female donor rapidly form a homogenous microglia population.
Time-lapse video capturing the rapid and homogeneous microglia phenotype acquisition upon thawing of cryopreserved female donor-derived ioMicroglia. 10 day time course.

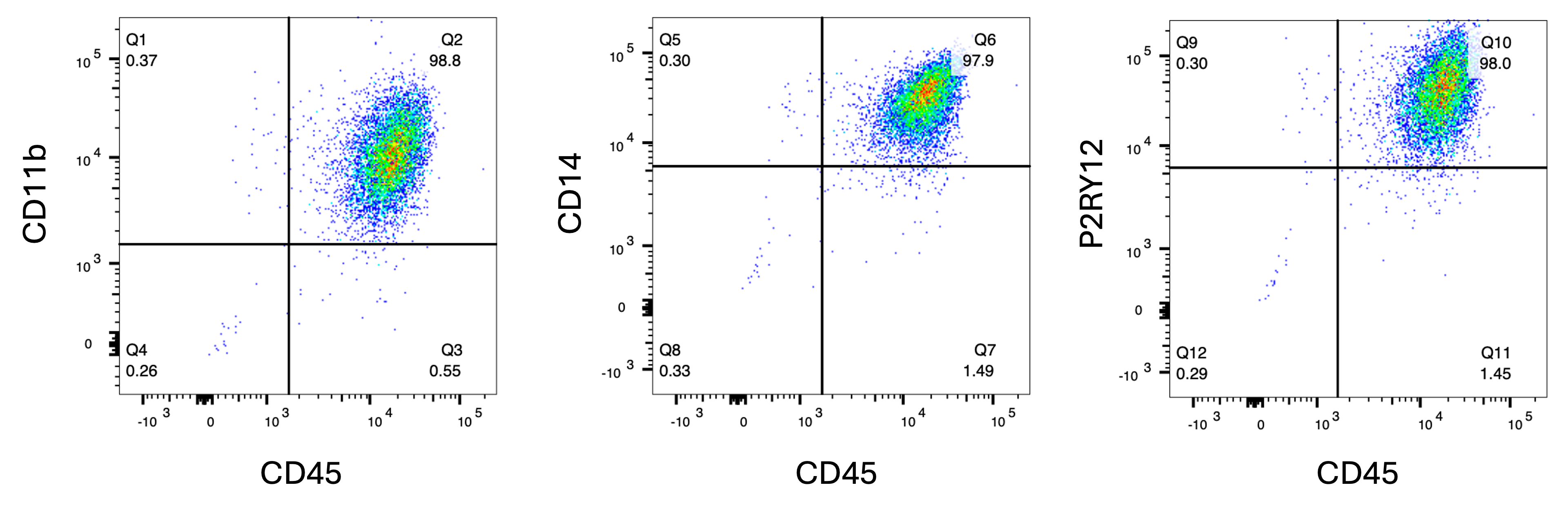
Flow cytometry analysis of female donor-derived ioMicroglia shows key phenotypic marker expression
Flow cytometry analysis of day 10 female donor-derived ioMicroglia shows key microglia marker expression of CD11b, CD45, CD14, and P2RY12 with a purity of above 97% for all these markers.
View the cell detachment protocol used to generate this data.

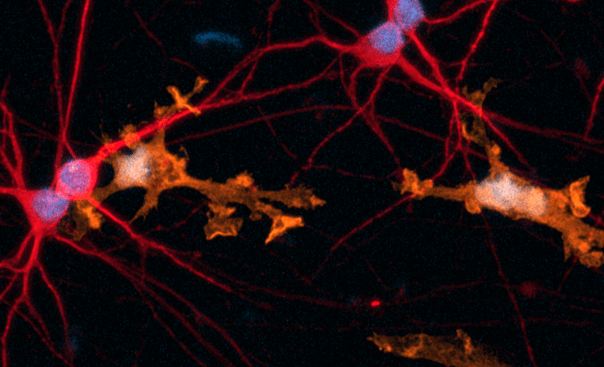
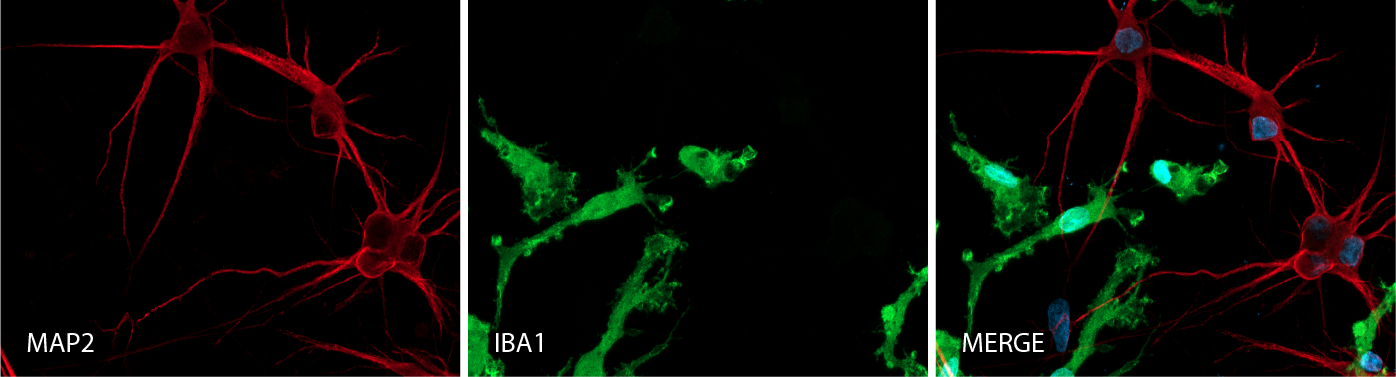
Key marker expression in female donor-derived ioMicroglia and ioGlutamatergic Neuron co-cultures
Immunofluorescent analysis at day 8 of the co-cultures shows expression of the microglia marker, IBA1 (green) and the pan-neuronal marker, MAP2 (red), as expected. Representative images taken at 10x magnification.
Female donor-derived ioMicroglia retain phagocytic function in co-culture with ioGlutamatergic Neurons
Representative video showing female-derived donor ioMicroglia in co-culture with ioGlutamatergic Neurons selectively phagocytosing pHrodo Red labelled Zymosan particles after 10 days in co-culture, without any observed adverse effects on neuron morphology. When female donor-derived ioMicroglia engulf these particles this causes the particles to fluoresce red, within the cells, due to the drop in pH in the phagolysosome. Live imaging was performed in 8-minute intervals over a time period of 1 hour and 36 minutes using the 3D Cell Explorer 96focus Nanolive Imaging system.
Female donor-derived ioMicroglia form co-cultures with ioGlutamatergic Neurons
ioGlutamatergic Neurons (io1001) were cultured to day 10 post-thaw. Female donor-derived ioMicroglia (io1029) cultured to either day 1 or day 10 post-thaw were added directly to day 10 ioGlutamatergic Neurons. The co-cultures were maintained for a further 6 days. Representative video showing that female donor-derived ioMicroglia form a stable co-culture with ioGlutamatergic Neurons. Live imaging was performed in 6.5-minute intervals over a time period of 3 hours and 31 minutes using the 3D Cell Explorer 96focus Nanolive Imaging system.

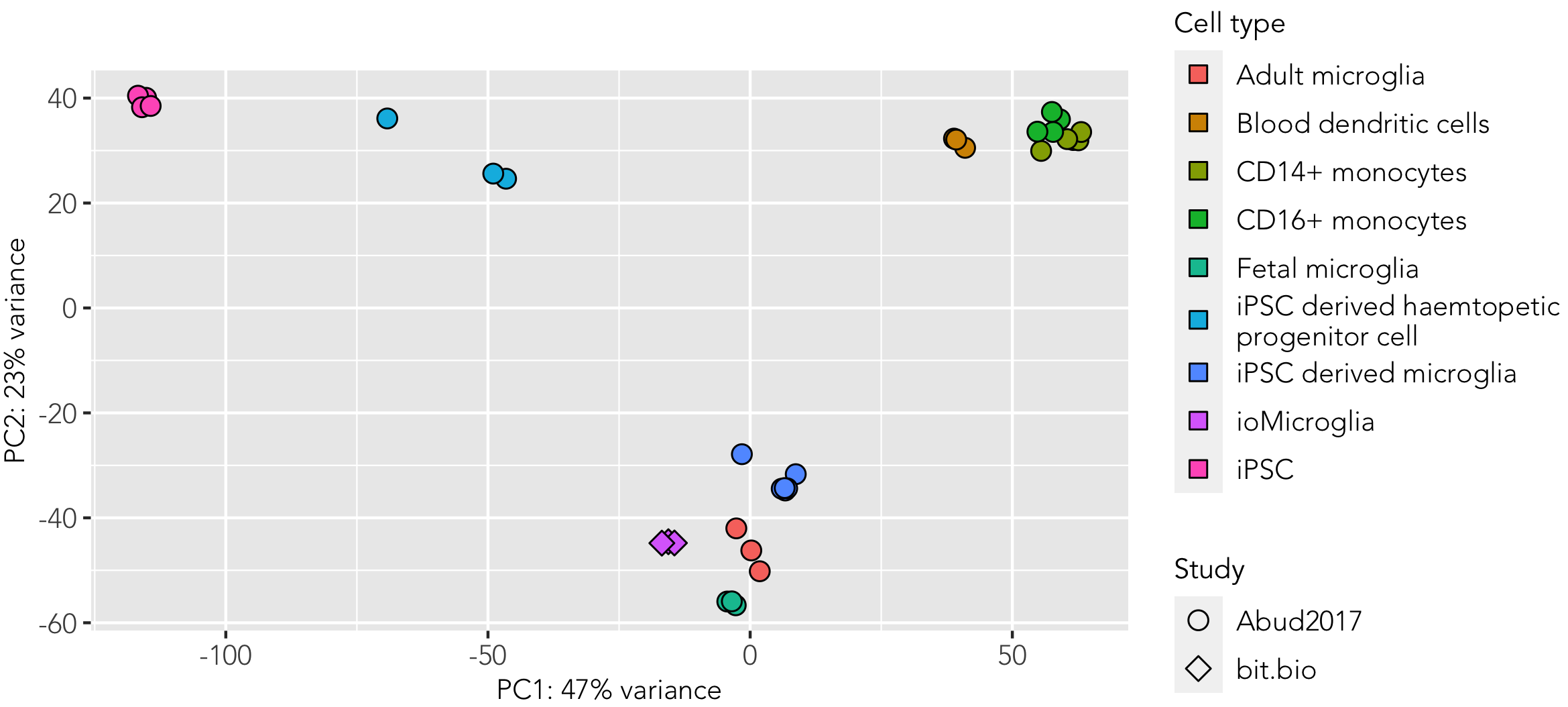
Whole transcriptome analysis demonstrates that female donor-derived ioMicroglia are highly similar to primary adult, foetal and other iPSC-derived microglia
Principal component analysis of bulk RNA sequencing data from female donor-derived ioMicroglia, integrated with sequencing data from Abud et al. (1) shows that these cells cluster closely to primary foetal and adult microglia data sets derived from this publication. Shapes represent the experiment from which data was obtained and colours represent the cell type.
(1) Abud E, et al., Neuron, 2018; 94(2): 278-293

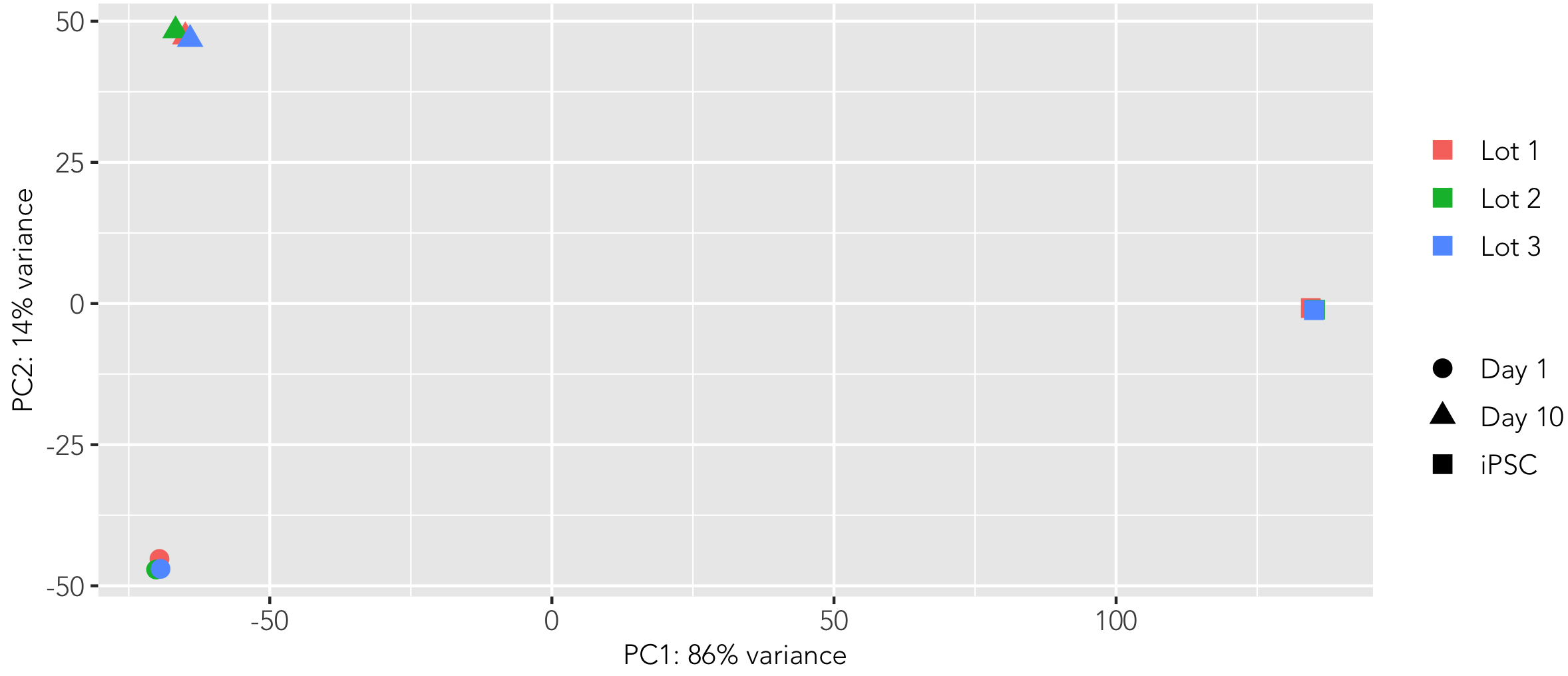
Whole transcriptome analysis demonstrates high lot-to-lot consistency of female donor-derived ioMicroglia
Bulk RNA sequencing analysis was performed on three independent lots of female donor-derived ioMicroglia at three different time points throughout the reprogramming protocol. Principal component analysis represents the variance in gene expression between the lots and shows the high consistency across each lot at each given time point. Populations of female donor-derived ioMicroglia with equivalent expression profiles can be generated consistently from every vial, allowing confidence in experimental reproducibility.

ioMicroglia are efficiently transfected with mRNA encoding GFP
ioMicroglia Male are efficiently transfected and show sustained long-term expression of mRNA encoding GFP. Cells were imaged throughout the experiment to assess transfection efficiency and evaluate potential cytotoxic effects of the transfection protocol. Day 4 images were captured prior to transfections on the same day.
Download the step-by-step protocol for lipid-based delivery of synthetic mRNA into ioMicroglia.
Vial limit exceeded
A maximum number of 20 vials applies. If you would like to order more than 20 vials, please contact us at orders@bit.bio.












Hoescht(blue)TUBB3(blue)_day4.png?width=604&name=bit.bio_ioGlutamatergic%20Neurons_60xMAP2(red)Hoescht(blue)TUBB3(blue)_day4.png)







_MAP2(R)_Tubb3(B)_Hoechst(B)_20x_merge-comp.jpg?width=604&name=Colour%20webinar%20with%20it-bio%20ioGlutamatergic%20Neurons_VGLUT2(G)_MAP2(R)_Tubb3(B)_Hoechst(B)_20x_merge-comp.jpg)