New Approach Methodologies (NAMs) are a set of human-relevant tools or techniques, from advanced cell culture to computational modelling, that align with the 3Rs principles to replace, refine or reduce the use of animals in experimentation. With the potential to become the new paradigm in drug development, the success of new approach methodologies relies on one critical factor: the consistent quality of their cellular components.
Human. Consistent. Reliable.
Complex models require a stable foundation. Without consistent cells, even the most sophisticated new approach methodologies risk producing variable results. ioCells, powered by opti-ox technology, provide deterministically programmed, consistent and defined human cells to support reproducible NAM performance. With ioCells, new approach methodologies can be transformed from experimental prototypes into robust tools for drug discovery.
Consistent inputs
ioCells offer a source of scalable, consistent human cells essential for NAMs development in research and drug testing.
Physiological relevance
Capture critical cell function to mimic human responses more accurately than animal models.
Scalable
Transitions NAMs from low-throughput research to industrial screening scale by providing consistent, bulk quantities of defined human cells.
ioCells in action
ioCells in action
Predictive human models: the spectrum of New Approach Methodologies (NAMs)

The NAMs ecosystem encompasses a broad spectrum of technologies, from cell culture-based approaches, bioengineering to computational modelling.
To better predict clinical outcomes, new approach methodologies are the space of integration of advanced cell culture systems with material engineering to simulate specific organ functions and tissue mechanics. The ioCells portfolio enables precise interrogation of disease mechanisms, CRISPR genomic and toxicity screenings, providing a platform for building powerful NAMs for tomorrow's medicines.
A paradigm shift in regulatory science: FDA's announcement to phase out animal testing

In a landmark decision, the FDA announced a plan to phase out animal testing requirements for monoclonal antibodies and new drugs. This move encourages the adoption of New Approach Methodologies (NAMs) to improve drug safety and efficiency, marking a pivotal moment for human-relevant science.
The FDA’s decision marks the dawn of a new era. By accepting proven alternatives to animal models, drug developers can now move toward safer, faster, and more ethical clinical trials without routine animal experimentation.
Industrialising cellular programming: Manufacturing human cells with unprecedented consistency
In this exclusive ISSCR presentation, Prof. Marius Wernig and Dr Mark Kotter discuss the evolution of cellular reprogramming from a biological paradigm to an industrial reality. Wernig outlines the foundational science of transcription factor-mediated cell identity, while Kotter explains how integrating this biology with opti-ox technology overcomes the variability of traditional methods. This approach enables the scalable, precise manufacturing of human cells needed to unlock the full potential of regenerative medicine.
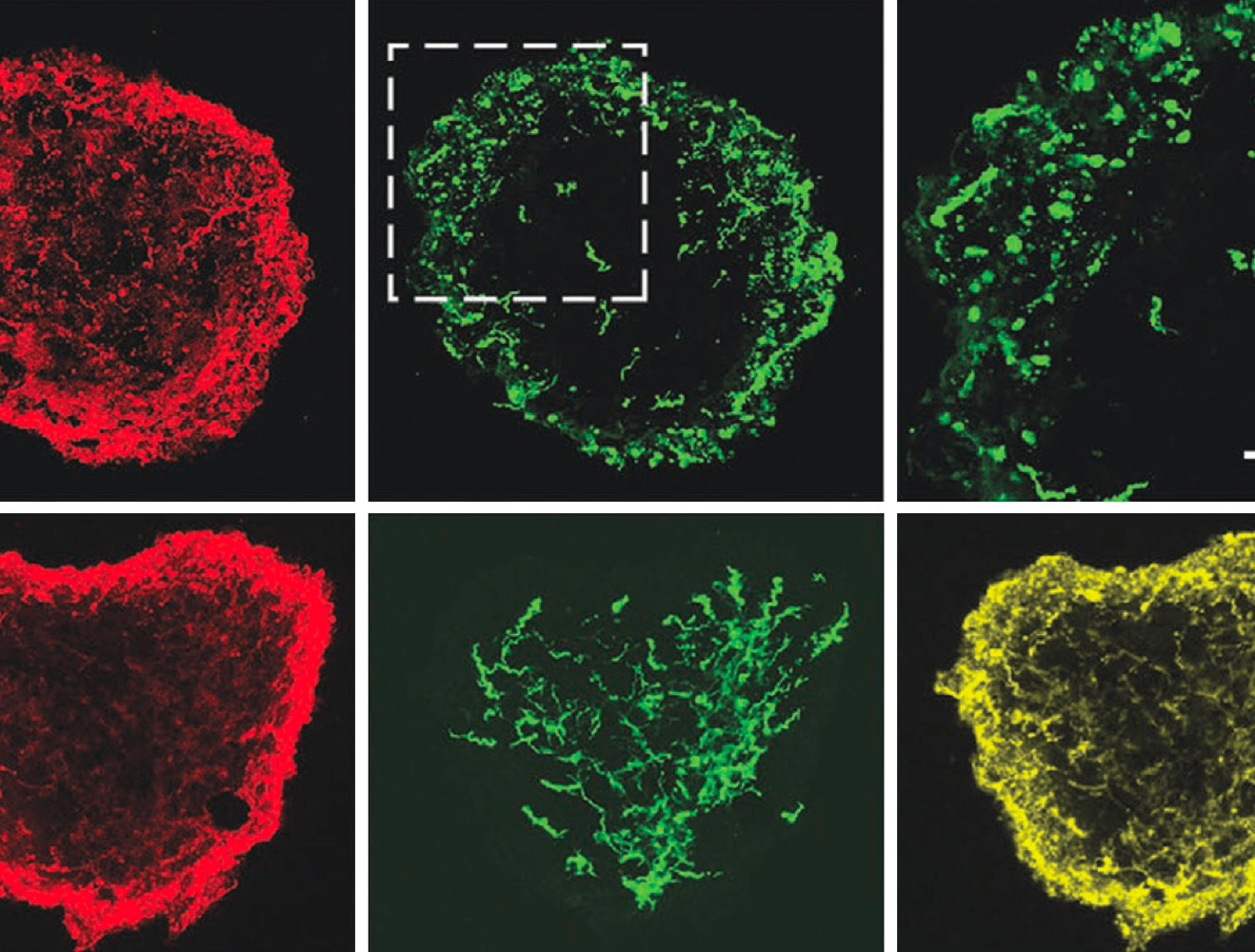
ioCells provide physiologically-relevant, functional models for studying cell-to-cell interactions

Immunocytochemistry staining of a tri-culture with ioGlutamatergic Neurons, ioAstrocytes and ioMicroglia. Cell cultures stained for MAP2 (neuron marker), Vimentin (astrocyte marker), and IBA1 (microglia marker), validating the integration of all three cell types.
Co-culturing multiple cell types allows researchers to mimic natural cell-to-cell and neuron-glial interactions. This tri-culture system provides a validated physiologically-relevant model to replicate neuroinflammatory and neurodegenerative pathways that isolated monocultures may miss.
Scalable 3D neuronal microtissues using ioGlutamatergic Neurons

Automated generation of human neuronal microtissues. ioGlutamatergic Neurons and iPSC-derived astrocytes were bioprinted using the Inventia RASTRUM™ platform to form 3D co-cultures in a CNS-mimetic hydrogel. The model supports high-throughput screening in 96- and 384-well formats, exhibiting mature phenotypes and extensive neurite outgrowth. Data courtesy of Inventia Life Science.
Printoids are 3D tissues engineered via bioprinting, where cells are deposited in precise, orderly spatial arrangements. Unlike self-assembling models, printoids allow for the standardised reconstruction of complex tissue interfaces. ioCells enable the generation of reproducible neuronal microtissues at scale.
Modelling neuromuscular interactions on-chip with ioSkeletal Myocytes and ioMotor Neurons

Developing advanced neuromuscular cultures. Dr Grace Cooper showcases a 28-day co-culture of ioMotor Neurons and ioSkeletal Myocytes. In this talk, she explores the development of physiologically relevant interactions in the NETRI DuaLink microfluidic system, creating an MEA-compatible model for studying synaptic development.
Organ-on-chip, or Microphysiological Systems (MPS), engineer physiology through geometry and flow. Cells are cultured in compartmentalised microfluidic devices to mimic tissue mechanics. These new approach methodologies model systemic interactions, such as long-range nerve signalling and axonal transport, that static plates cannot capture.
.webp?width=2400&height=600&name=Website%20in%20tabs%20image%20-%20landscape%20April%2026%20(1200%20x%20300%20px).webp)
With a defined input and an advanced architecture (NAMs), it is possible to generate a reliable, reproducible output to transform NAMs from experimental concepts to into robust industrial tools for drug discovery and toxicity testing.
Download the poster to see how ioCells are being incorporated into a variety of NAMs to support the transition to human-specific research.
What scientists say about ioCells

Dr Mariangela Iovino
Senior Group Leader | Biology Discovery | Charles River
“Our major surprise when we first used the ioGlutamatergic Neurons was that after thawing the cells in 384-well format, we could see immediately after 2 days a nice neuronal network, and there was no well to well variability within the same plate. This made our assay quite robust.”

Dr Koby Baranes
Research Associate | University of Cambridge
"These cells provide a reliable and pure source of glutamatergic neurons, resembling primary human ones. They are ready-to-use which makes it much more easy for tissue culture work and for reproducible results.”

Dr Shushant Jain
Group Leader | In Vitro Biology | Charles River, 2021
"These cells enable us to move rapidly as from the moment of plating within 4-7 days we have mature and functional neurons."

Dr Michael Duchen
Professor of Physiology | University College London
“We can now start asking questions that, ten years ago, we didn’t know how to answer,” Duchen reflects. “If you have a really good disease model, then the only limit is your imagination.”

Dr Rick Klausner
Chief Executive Officer | Lyell Immunopharma
"bit.bio is based on beautiful science. The company’s technology has the potential to bring the long-awaited precision and reliability of engineering to the application of stem cells. bit.bio’s approach represents a paradigm shift in biology that will enable a new generation of cell therapies, improving the lives of millions."

Dr Marijn Vlaming
Head of Biology - Beerse & Leiden | Charles River
"Using more relevant cell models such as ioCells from bit.bio helps us to test safety and toxicity in vitro, in parallel with efficacy, and as such we will be able to identify the safe or unsafe drug candidates very early in the drug discovery process"

Emma Jones, PhD
Senior Scientist | Neurobiology Medicines Discovery Catapult
"More physiologically relevant disease models will permit better and more predictable drug discovery, and will therefore improve the translation of medicines to the clinic"

Jeremy Krohn
PhD Candidate | DZNE / Charité University of Medicine
"We used bit.bio human ioAstrocytes in our experiments to compare with - and step-wise replace - animal derived cells. The human cells were straightforward to establish, showed high reproducibility, and performed well in calcium imaging experiment."
Recommended resources
3D printing: Human cerebral cortex cells
In this study, researchers developed a lipid-bilayer-supported technique to 3D print ioGlutamatergic Neurons in soft Matrigel. This approach enables precise spatial control without synthetic scaffolds, revealing how initial cell positioning influences human cortical development.

Decision-making in drug development
Predictive validity in drug discovery involves a careful balance between model complexity, scalability, and physiological relevance. Research is moving from using simple assays and animal models to building complex human-relevant systems. Discover how human iPSC-derived cells provide a scalable, physiologically-relevant source of cellular building blocks needed for better preclinical outcomes.

WC13 2025: Global momentum for human-relevant science
At the 13th World Congress on Alternatives and Animal Use, the consensus was clear: the shift to animal-free science is inevitable. Discover key insights on regulatory momentum, the rise of AI in toxicology, and the global push for human-relevant NAMs as alternatives to animal models.
Contact us
Developing robust New Approach Methodologies (NAMs) requires expert support to find the perfect building blocks for your new approach method. Our scientific team is ready to discuss your specific assay design, from co-culture protocols to media compatibility, ensuring your advanced model starts with a consistent foundation.
.webp)
bit.bio is proud to be part of The 3Rs Collaborative’s (@NA3RsC) Microphysiological Systems (MPS) initiative. The MPS initiative aims to increase industry adoption and regulatory acceptance of MPS technologies, to refine, reduce, and replace the use of research animals.
Frequently Asked Questions (FAQs)
1. What are New Approach Methodologies (NAMs)?
NAMs are a set of innovative, human-relevant tools, ranging from simple to advanced cell culture systems (like spheroids, organoids or microphysiological systems) to computational modelling, designed to replace or complement in vivo animal studies. They aim to better capture human biology in vitro to more accurately predict drug and new biologics efficacy and toxicity.
2. Why is cellular consistency critical for NAMs?
However complex the new approach method (from mono to multi-cell cultures, spheroids or organ-on-chip models), they all rely on a stable cell foundation to produce reliable data. If the cellular input varies from lot to lot, the model's advanced architecture cannot compensate, resulting in unreliable results. ioCells provide defined, consistent human cell inputs, ensuring that the data generated reflects true biological response rather than experimental variability.
3. Why are ioCells ideal for NAMs?
NAMs require high reproducibility to be useful in drug discovery pipelines. ioCells are generated using opti-ox technology, ensuring lot-to-lot consistency. This eliminates the variability associated with primary animal cells or iPSC-derived cells generated via directed differentiation, providing a reliable foundation for human-based culture systems.
4. What types of models are considered new approach methodologies (NAMs)?
NAMs encompass a spectrum of human-relevant in vitro systems ranging in complexity. At the foundational level, they include 2D mono or multi-cell cultures that establish cell-to-cell interactions between cell types, mimicking physiological context in vitro. Moving into 3D, spheroids and organoids recapitulate tissue architecture and density. At the most advanced end, bioengineered solutions such as printoids (bioprinted tissues) and organ-on-a-chip microfluidic devices introduce fluid flow and physical geometry to model systemic physiology.
5. How do NAMs serve as effective alternatives to animal models in drug discovery?
Animal models often fail to predict clinical outcomes due to biological differences between species. As predictive alternatives to animal models, new approach methodologies utilise human cells to model in vitro normal or disease biology and assess drug response with greater accuracy. This allows drug discovery teams to screen candidates for efficacy and toxicity in a human-relevant context across all preclinical stages, significantly de-risking the pipeline and preventing costly late-stage trial failures.
Ready to browse?
Explore our full portfolio of human iPSC-derived ioCells. From neurons and glia to muscle and immune cell types, our products are consistent and defined, supporting reliable data generation for applications in disease research and drug discovery.