Consistent, scalable, and highly pure sensory neurons with a defined nociceptor identity
Powered by opti-ox
Powered by opti-ox
Nociceptive sensory neurons are specialised neurons of the peripheral nervous system responsible for detecting painful, thermal, and chemical stimuli. These neurons in the dorsal root ganglia form the basis of the body's protective sensory network, signalling potential or actual tissue damage to the central nervous system. Disturbance of these pathways is a primary driver of chronic pain, a debilitating condition affecting nearly a third of adults for which current therapies are often inadequate.
Researchers often rely on sensory neuron models with significant drawbacks: animal models translate poorly to humans, immortalised cell lines lack physiological relevance, and directed differentiation methods are slow and can yield heterogeneous cell populations (contaminated by proliferating non-neuronal cells), compromising assay reliability.
ioSensory Neurons provide scientists a defined, highly pure source of human iPSC-derived sensory neurons with a defined nociceptive identity.
This time-lapse captures the rapid maturation of ioSensory Neurons, human iPSC-derived sensory neurons, over a 14-day time course. Upon thawing, the cells differentiate to quickly establish a homogeneous neuronal network. By day 7, they form a highly pure (>99%) sensory neuronal population with a defined nociceptor identity, providing scientists with a consistent, scalable model.

ioSensory Neurons are suitable for automated patch clamp analysis, and achieved 100% capture and >80% seal rates on the Patchliner from Nanion Technologies. Recordings confirmed TTX sensitive and TTX insensitive sodium channel currents, ligand-gated responses to GABA, glutamate, and P2X, in addition to heat-activated currents, and action potential firing under current clamp.
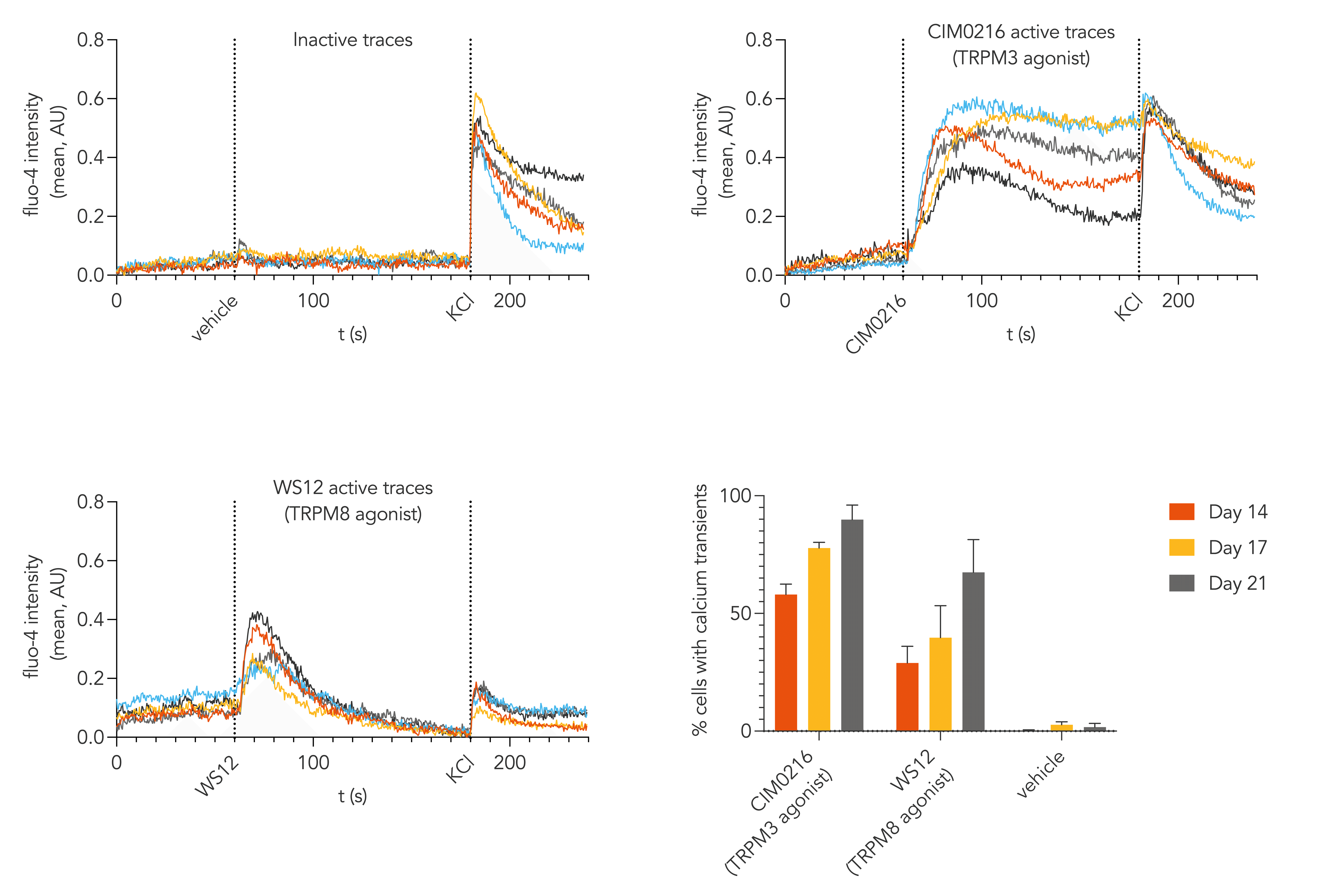
Calcium imaging confirms that ioSensory Neurons display robust nociceptor functionality, with 90% of cells responding to TRPM3 agonist CIM0216 and 67% to TRPM8 agonist WS12 when cultured for 21 days which were enhanced using specially optimised media for TRP channels.
At the Human Cell Forum 2025, Dr Ryan Jones, Cardiff University, presented a novel approach using ioSensory Neurons to model the bone-nociceptor interface. While traditional human osteoarthritis in vitro models capture inflammation, they do not include a nociceptive element. This model fills a gap, enabling researchers to investigate the molecular mechanisms and excitability changes driving chronic pain in osteoarthritis.
In this video, our scientist takes you through the step-by-step process of how to thaw, seed and culture human iPSC-derived sensory neurons (ioSensory Neurons).
.jpg?width=713&height=549&name=Ryan%20jones%20headshot%20(2).jpg)
Dr Ryan Jones
Postdoctoral Research Associate | Cardiff University

Looking to add an inflammatory component to your CNS studies?
Move into a multi-cellular model using different cell types to mimic peripheral cell interaction
ioMicroglia Male
ioOligodendrocyte-like cells
ioAstrocytes

Build your custom disease model or reporter line to pair with wild-type ioSensory Neurons as the genetically matched control.
Throughout the custom process, our experts will bring your project to life, and be on hand to support you with any technical queries.
Start the conversation today


ioSensory Neurons are highly pure, defined and consistent human iPSC-derived sensory neurons, a great companion for your CNS model.
Combine them with other neuronal and glia ioCells to generate complex multi-cell cultures.
ioGlutamatergic Neurons
ioMicroglia
ioAstrocytes
ioOligodendrocyte-like cells

Access 20 neuronal disease models and 4 microglia disease models with a single co-culture protocol
View the co-culture protocol
Explore ioGlutamatergic Neuron Disease Models
Explore ioMicroglia Disease Models
Oosterveen et al.
bit.bio
2024
Davenport A, Frolov T & Kotter M
Drug Discovery World
2020
Innovation showcase talk at ISSCR
Marius Wernig MD, PhD | Stanford
Mark Kotter, MD, PhD | bit.bio
Dr Ryan Jones | Research Associate | Cardiff University
Human Cell Forum 2025
Session 1 Track 2 | From cells to systems: Building human iPSC-derived models of pain, neuromuscular junctions, and glial dynamics
Dr Shushant Jain | Group Leader In Vitro Biology | Charles River
Interview at SLAS
Dr Mariangela Iovino | Group Leader | Charles River
Dr Tony Oosterveen | Senior Scientist | bit.bio
Dr Ania Wilczynska | Head of Computational Genomics | Non-Clinical | bit.bio
Prof Roger Pedersen | Adjunct Professor and Senior Research Scientist at Stanford University
Dr Thomas Moreau | Director of Cell Biology Research | bit.bio
Nociceptive sensory neurons are specialised cells of the peripheral nervous system (PNS) responsible for detecting potentially damaging stimuli, including extreme temperatures, mechanical stress, and chemical irritants. As the body's primary protective network, these neurons transmit pain signals to the central nervous system (CNS). Damage to sensory neuron networks is thought to be a key driver of chronic pain conditions.
ioSensory Neurons are human iPSC-derived sensory neurons that help to address the poor translation of animal models and the heterogeneity of conventional directed differentiation iPSC models by providing a defined, >99% pure nociceptor population. Powered by opti-ox technology, this consistent human model showcases none of the contamination from non-neuronal cells often seen in standard protocols, supporting assay reliability and scalability.
ioSensory Neurons rapidly differentiate to establish a homogeneous neuronal network, achieving a highly pure phenotype with defined nociceptor identity by day 7 post-thaw. This rapid maturation allows scientists to bypass lengthy differentiation protocols and access a consistent, scalable source of human iPSC-derived sensory neurons for drug discovery workflows.
Validated in automated patch clamp, ioSensory Neurons display robust physiological functionality, including TTX sensitive and TTX resistant voltage-gated sodium currents and ligand-gated responses to GABA and glutamate. Calcium imaging analysis demonstrated that ioSensory Neurons have a high response rate to TRPM3 and TRPM8 agonists when cultured in optimised media.
ioSensory Neurons enable the creation of complex in vitro pain models, such as the bone-nociceptor interface to study osteoarthritis. Unlike traditional osteoarthritis in vitro models that only capture inflammation, this human nociceptor model allows scientists to study the specific molecular mechanisms and excitability changes that drive chronic pain.