Access a toolkit of functional, consistent inhibitory GABAergic neurons in to study neurodegenerative diseases
Powered by opti-ox
Powered by opti-ox
GABAergic neurons are the major inhibitory neurons of the central nervous system (CNS), playing a critical role in balancing excitatory signals. This excitatory-inhibitory balance is fundamental to healthy brain function by regulating neuronal firing and network synchronisation. Consequently, dysfunction in GABAergic signalling is strongly associated with neurological and psychiatric disorders, including Alzheimer’s disease, epilepsy, autism spectrum disorder (ASD), and schizophrenia.
Developing therapeutics for these conditions requires reliable, human-relevant models; however, current approaches often struggle to generate inhibitory interneurons with sufficient purity and consistency for high-throughput screening.
ioGABAergic Neurons provide a highly pure (>99%) population of human iPSC-derived inhibitory neurons powered by opti-ox technology. Ready for experimentation within just 4 days of thawing, these cells express classical GABAergic neuron markers and, when in co-culture with glutamatergic neurons, help to modulate neuronal network activity, making them ideal for precisely studying excitatory/inhibitory (E/I) balance and disease mechanisms.
ioGABAergic Neurons rapidly acquire a homogeneous phenotype, as captured in this video time course. Powered by opti-ox technology, these cells quickly mature into a defined, >99% pure population of GABAergic inhibitory interneurons. Unlike the heterogeneous populations generated by traditional directed differentiation methods, ioGABAergic Neurons provide scientists with a highly pure, human in vitro model to precisely study changes in inhibitory/excitatory balance.
Calcium imaging reveals the rapid acquisition of functional activity in ioGABAergic Neurons. As captured in this video, the cells demonstrate robust spontaneous firing at day 16 post-revival. This early functional maturation enables scientists to accelerate timelines for assay development and neuronal network analysis.

Tri-cultures of ioGlutamatergic Neurons, ioGABAergic Neurons, and astrocytes form functional neuronal networks and modulate network activity. MEA analysis reveals that systematically increasing the ratio of ioGABAergic Neurons reduces the synchronised network activity driven by excitatory neurons, as expected. The absence of bursting in GABA-only controls confirms the high purity of the inhibitory population. These networks achieve stable spontaneous activity by DIV 25, which is maintained through DIV 64. Experiments performed by Charles River Laboratories on the Axion Maestro Pro MEA platform.

MEA analysis on the Axion Maestro Pro platform demonstrates the specific inhibitory effect of ioGABAergic Neurons in tri-cultures with ioGlutamatergic Neurons and astrocytes. Treatment with diazepam—a GABAA receptor positive allosteric modulator— reduces network activity in tri-cultures but shows no impact on the control co-cultures. The representative raster plots and network activity quantification demonstrate a dose-dependent decrease in spikes per network burst, confirming the system as a sensitive, robust in vitro model for GABAergic modulation in drug discovery.
In this case study, Rodney A. Bowling Jr., PhD, founder and CSO of To Cure a Rose, shares insights on their need for consistent human neurons that express a target gene. Easy access to gene expression data on ioCells assisted them in selecting the right cells, ioGABAergic Neurons, and discovering not just one, but two promising ASO candidates.
The defined, consistent nature of the cells enabled the successful assessment of 57 antisense oligonucleotides (ASOs), identifying two lead candidates with >90% knockdown efficacy.

ioGABAergic Neurons Alzheimer’s disease models carry a genetically engineered homozygous and heterozygous APP V717I (London) mutation, recapitulate the expected Alzheimer’s disease phenotype of Amyloid beta (Aβ) peptide release in an MSD assay over a 30-day time course. These disease model cells enable reliable investigations APP mutant protein when paired with the genetically matched wild-type ioGABAergic Neurons.
In this video, our scientist takes you through the step-by-step process of how to thaw, seed and culture ioGABAergic Neurons.

Rodney A. Bowling Jr., Ph.D.
Co-Founder & Chief Scientific Officer | Everlum Bio
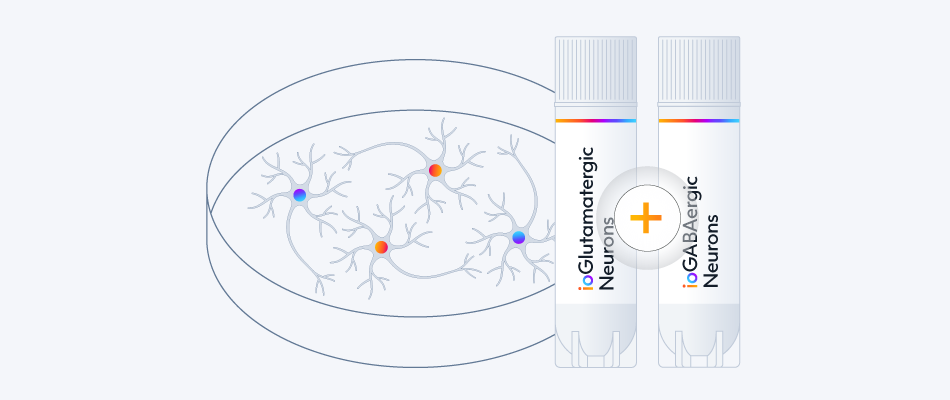

Build neuronal co- and tri- cultures that more accurately represent the complexity of the human brain. Glutamatergic and GABAergic neurons are crucial to help maintain stable ratios of excitatory and inhibitory populations throughout your MEA experiments.
Explore ioGlutamatergic Neurons
Explore ioGlutamatergic Neuron Disease Models


Investigate the impact of the APP V717I (London) mutation on Alzheimer's disease pathology with a functional, rapidly maturing, and disease-relevant system.
ioGABAergic Neurons engineered with the APP V717I (London) mutation demonstrate disease-related phenotype of an increased ratio of A𝛽42:40, as observed in Alzheimer’s disease, when compared to wild-type ioGABAergic Neurons, their genetically matched control.
View the data on the product pages
ioGABAergic Neurons APP V717I/V717I
ioGABAergic Neurons APP V717I/WT
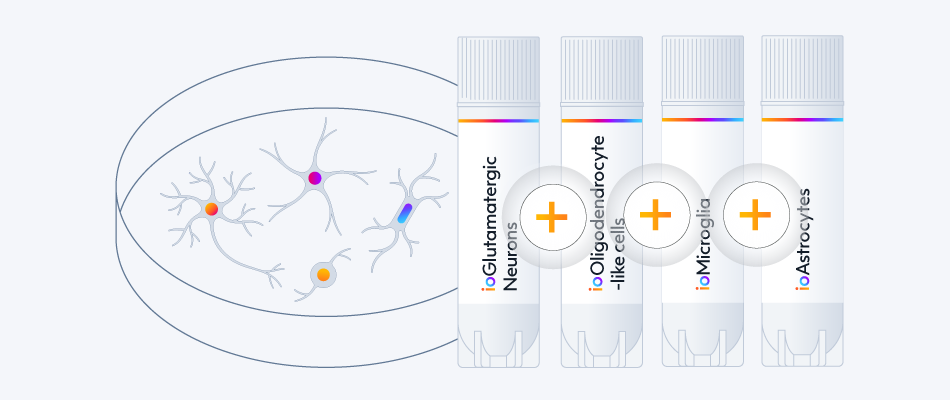

ioGABAergic Neurons are highly pure, defined and consistent, a great companion for your CNS model.
Combine them with other neuronal and glia ioCells to generate complex multi-cell cultures.
ioGlutamatergic Neurons
ioMicroglia
ioAstrocytes
ioOligodendrocyte-like cells
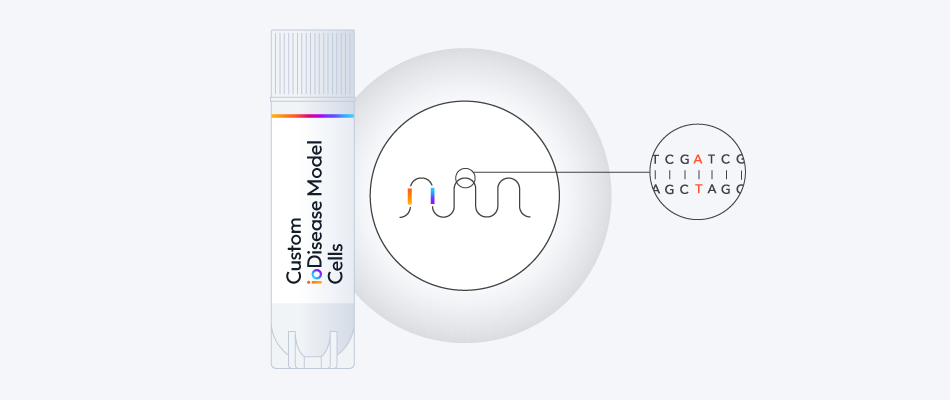

Build your custom disease model or reporter line to pair with wild-type ioGABAergic Neurons as the genetically matched control.
Throughout the custom process, our experts will bring your project to life, and be on hand to support you with any technical queries.
Start the conversation today
bit.bio
Smith, et al.
bit.bio
2024
Davenport A, Frolov T & Kotter M
Drug Discovery World
2020
Guerrisi S, et al.
BioRxiv
2026
Using ioGlutamatergic Neurons, ioGABAergic Neurons, ioMicroglia and ioMicroglia TREM2 R47H/R47H
Innovation showcase talk at ISSCR
Marius Wernig MD, PhD | Stanford
Mark Kotter, MD, PhD | bit.bio
Dr Deepak Srivastava | King’s College London
Dr Mariangela Iovino | Group Leader | Charles River
Dr Tony Oosterveen | Senior Scientist | bit.bio
Prof Roger Pedersen | Adjunct Professor and Senior Research Scientist at Stanford University
Dr Thomas Moreau | Director of Cell Biology Research | bit.bio
Dr Brian Gill, MD | Assistant Professor of Neurological Surgery| Columbia University Irving Medical Center
Dr Tony Oosterveen | Principal Scientist and CNS Lead, Neurobiology | bit.bio
GABAergic neurons are the primary inhibitory neurons of the central nervous system (CNS), critical for maintaining the excitatory-inhibitory (E/I) balance to carefully modulate neuronal network activity. This excitatory-inhibitory balance is fundamental to healthy brain function, regulating spontaneous firing and network synchronisation. Consequently, dysfunction in GABAergic signalling is strongly associated with neurological disorders like Alzheimer’s disease, epilepsy, autism spectrum disorder (ASD), and schizophrenia.
ioGABAergic Neurons address the heterogeneity obtained with traditional directed differentiation protocols by providing a >99% pure population of inhibitory interneurons. Powered by opti-ox technology, these human iPSC-derived GABAergic neurons rapidly acquire a homogeneous and functional phenotype, eliminating the noise and variability that affects CNS models from other GABAergic neuron models.
ioGABAergic Neurons are ready for experimentation within 4 days post-thaw and display robust, spontaneous functional activity by day 16 when assess by calcium imaging. This rapid maturation allows scientists to avoid months-long differentiation protocols, significantly accelerating assay development for neuronal network analysis.
In co-culture with excitatory neurons, ioGABAergic Neurons actively modulate network dynamics, effectively dampening synchronised bursting to establish a physiological excitatory-inhibitory (E/I) balance. Functional validation using microelectrode array (MEA) analysis confirms that ioGABAergic Neurons respond to GABA modulating compounds, such as diazepam, which enhances their inhibitory effect in a dose-dependent manner.
ioGABAergic Neurons engineered with the APP V717I (London) mutation recapitulate Alzheimer’s disease, exhibit the expected disease-specific phenotypes such as an increase in the Aβ42:40 ratio. This genetically matched model mirrors the amyloidogenic shift found in disease, providing a robust, human-relevant platform for screening therapeutics against wild-type controls.