Access consistent, co-culture ready human astrocytes
Powered by opti-ox
Powered by opti-ox
Astrocytes are glial cells, essential for maintaining homeostasis and defence within the central nervous system (CNS). Astrocytes form complex networks to regulate energy metabolism, maintain the blood-brain barrier, and ensure synapse formation and maturation. Dysfunction of these cells is increasingly implicated in the progression of CNS disorders and neurodegenerative diseases.
Despite their importance, the precise role of astrocytes in disease remains poorly understood, and replicating their complex functions in vitro is a challenge. Moreover, therapeutic development has historically focused on neurons, often neglecting the significant, and sometimes unintentional, effects of drug compounds on astrocytes. The lack of pure, functional, and scalable human astrocyte models has limited the development of robust screening pipelines to investigate these effects. ioAstrocytes provide scientists with a reliable and scalable source of human iPSC-derived astrocytes that mimic essential functions such as inflammatory responses, protein clearance, and neuronal support.
Modelling astrocytic phagocytosis, a key process in neuroinflammation and debris clearance, is often challenging. Primary astrocytes show high lot-to-lot variability, compromising the reproducibility of functional assays and making it difficult to obtain consistent results.
In this video, ioAstrocytes readily phagocytose Red S. aureus Bioparticles®, giving scientists confidence in a reliable human model to quantify phagocytic activity. At the start of the video, the particles are located outside the cells and due to the neutral pH of the media are non-fluorescent, but when phagocytised, they are exposed to the acidic environments of intracellular organelles and fluoresce bright red.

ioAstrocytes provide a robust platform to model neuroinflammation in vitro. When treated with proinflammatory stimuli, astrocytes secret a range of relevant cytokines, including the strong release of Interleukin 6 (IL-6).

Jeremy Krohn (DZNE) emphasises the critical role of astrocytes as mediators of brain function and their impact on therapeutic response. Using calcium imaging as a functional readout, the research demonstrates how human iPSC-derived ioAstrocytes and neuron-astrocyte co-cultures provide a superior alternative to animal models. By capturing spontaneous and evoked activity in human-relevant systems, the data highlights how ioAstrocytes respond uniquely to drugs and disease stressors, ensuring more accurate translation from the lab to the clinic.

Our scientists have developed a complete co-culture protocol, which includes simple step-by-step instructions for cell culture, recipes for media and information on required equipment and reagents, for phenotypic readouts like immunocytochemistry staining.

When challenged with three distinct proinflammatory cocktails, ioAstrocytes exhibit a robust transition to a reactive state, characterised by the significant induction of cytokines such as IL-6. While the presence of IFN-gamma and IL-1beta in the initial stimuli leads to high detection signals, the overall secretory profile confirms a physiologically relevant neuroinflammatory response.
In this video, our scientist takes you through the step-by-step process of how to thaw, seed and culture ioAstrocytes.

Jeremy Krohn
PhD Candidate | DZNE / Charité University of Medicine
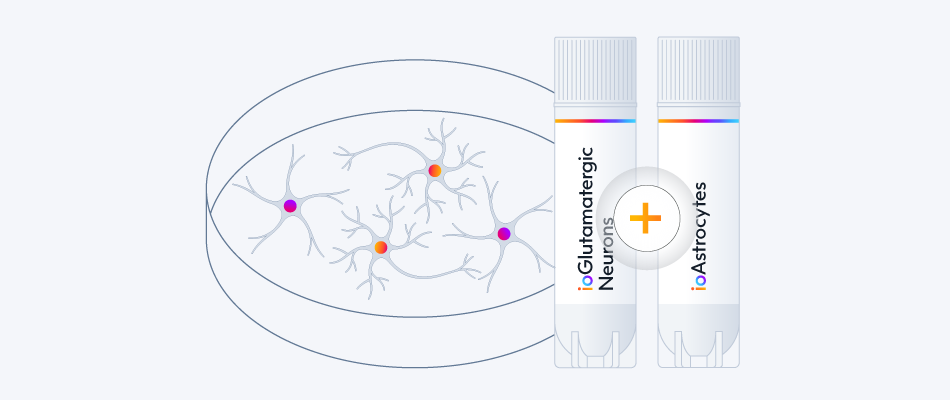

Access 20 neuronal disease models with a single co-culture protocol.
View the co-culture protocol
Explore ioGlutamatergic Neuron Disease Models
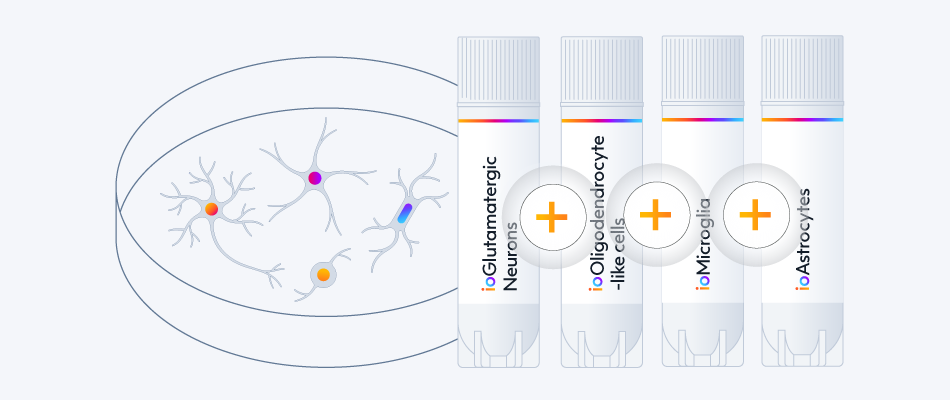

ioAstrocytes are highly pure, defined and consistent, a great companion for your CNS model.
Combine them with other neuronal and glia ioCells to generate complex multi-cell cultures.
ioGlutamatergic Neurons
ioMicroglia
ioOligodendrocyte-like cells


Interested in optimising your neuroinflammation studies?
Move into a multi-cellular model using different cell types:
ioMicroglia Male
ioGlutamatergic Neurons
ioOligodendrocyte-like cells
Tailor experimental conditions to answer your scientific question by including ioDisease Model Cells with disease-related mutations.


Build your custom disease model into the ioAstrocytes wild type. Use the genetically matched control in your experiments to make true comparisons in your data, being confident that even subtle variances in your data are attributable to your mutation of interest.
Start the conversation today
bit.bio
Davenport A, Frolov T & Kotter M
Drug Discovery World
2020
Jeremy Krohn | PhD Candidate | DZNE / Charité University of Medicine
Human Cell Forum 2025
Session 1 Track 2 | From cells to systems: Building human iPSC-derived models of pain, neuromuscular junctions, and glial dynamics
Dr Brian Gill, MD | Assistant Professor of Neurological Surgery| Columbia University Irving Medical Center
Dr Tony Oosterveen | Principal Scientist and CNS Lead, Neurobiology | bit.bio
Astrocytes are glial cells that regulate metabolism, maintain the blood-brain barrier, and facilitate synapse maturation. Since the dysfunction of astrocyte networks is often implicated in neurodegenerative conditions, accessing consistent human iPSC-derived astrocytes is essential for experimental workflows.
Research utilising primary astrocytes is often hindered by high lot-to-lot variability, which compromises the reliability of experimental data. Human iPSC-derived ioAstrocytes bypass this challenge by providing a scalable and defined source of human cells that exhibit a consistent phenotype, ensuring that experimental data is reproducible over time.
While drug discovery has historically prioritised neurons, there is a growing recognition that the inclusion of glial cells is critical to study the effect of drug compounds on astrocytes. Using human iPSC-derived astrocytes allows scientists to use a human model, revealing how drugs interact with glial cells and ensuring that therapeutic candidates are evaluated in a system that better reflects human biology.
Human iPSC-derived astrocytes, ioAstrocytes, offer a robust platform for investigating neuroinflammatory pathways and immune responses. These cells readily phagocytose bioparticles and secrete cytokines like IL-6 in response to proinflammatory stimuli, enabling the precise quantification of inflammatory states in vitro.
To better mimic the complexity of the brain, ioAstrocytes are fully compatible with co-culture workflows involving neurons, such as ioGlutamatergic Neurons. These multi-cellular cultures facilitate the study of critical neuroglial interactions in a relevant human model.
Kuijlaars, Jacobine, et al. "Sustained synchronized neuronal network activity in a human astrocyte co-culture system." Scientific Reports, 6, 36529, 2016, https://doi.org/10.1038/srep36529
Taga, Arens, et al. "Role of Human-Induced Pluripotent Stem Cell-Derived Spinal Cord Astrocytes in the Functional Maturation of Motor Neurons in a Multielectrode Array System." Stem Cells Transl Med, 8(12):1272-1285, 2019, doi: 10.1002/sctm.19-0147
Thiry, Louise, et al. "Characterization of Human iPSC-derived Spinal Motor Neurons by Single-cell RNA Sequencing." Neuroscience, Volume 450, P57-70, DOI: 10.1016/j.neuroscience.2020.04.041
Thiry, Louise, et al. "Optimization of Long-Term Human iPSC-Derived Spinal Motor Neuron Culture Using a Dendritic Polyglycerol Amine-Based Substrate." ASN Neuro, 14(1). https://doi.org/10.1177/17590914211073381