cat no | io1112 Early Access
GFP ioMotor Neurons
Human iPSC-derived motor neurons constitutively expressing GFP
- Cryopreserved human iPSC-derived cells powered by opti-ox that are ready for experiments in days
- Motor Neurons constitutively expressing GFP, easy to track in multi-cell cultures, ideal for live-cell imaging
- Co-culture compatible with ioSkeletal Myocytes and astrocytes
Human iPSC-derived motor neurons constitutively expressing GFP
Flow cytometry analysis of GFP expression at day 1, day 11 and day 21
Flow cytometry analysis demonstrating GFP expression in >99% of cells for GFP ioMotor Neurons at Day 1 (centre-left) and Day 11 (centre-right). No GFP expression seen in wild-type ioMotor Neurons (left). At day 21, the percentage of cells expressing GFP has not decreased, indicating there is no silencing of the reporter gene (right).
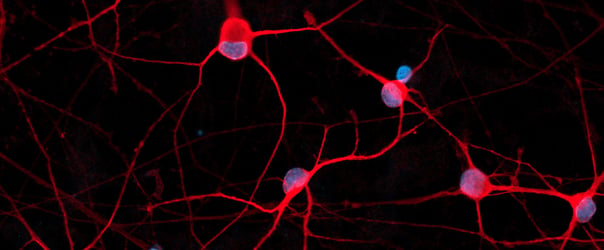
GFP ioMotor Neurons exhibit comparable expression of neuron-specific markers to the wild-type control
Immunofluorescent staining 11 days post-revival shows similar homogenous expression of cholinergic marker (ChAT), and motor neuron marker (ISL2) in both GFP ioMotor Neurons and the wild-type control.
The GFP signal is visible exclusively in GFP ioMotor Neurons and absent in the wild-type control.
GFP ioMotor Neurons exhibit comparable expression of neuron-specific markers to the wild-type control
Immunofluorescent staining 11 days post-revival shows similar homogenous expression of pan-neuronal marker (MAP2), cholinergic marker (VAChT), and motor neuron marker (HB9) in both GFP ioMotor Neurons and the wild-type control.
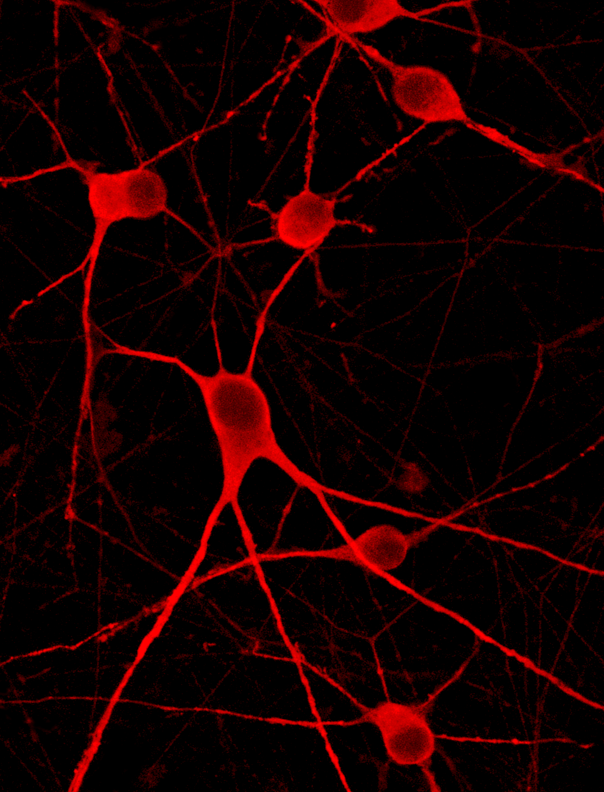
GFP ioMotor Neurons form a homogenous neuronal network by day 4, without clumping
GFP ioMotor Neurons mature rapidly, show motor neuron morphology and form structural neuronal networks over 14 days with no sign of clumping of coalescing of cell bodies. Day 1 to 14 post thawing. 400 µm scale bar.
GFP ioMotor Neurons demonstrate gene expression of neuronal-specific and motor-neuron-specific markers following deterministic programming
Gene expression analysis demonstrates that GFP ioMotor Neurons (GFP) and wild-type ioMotor Neurons (WT) lack the expression of pluripotency marker OCT4 (POU5F1) at day 11, while robustly expressing pan-neuronal (TUBB3 and MAP2), motor-neuron-specific (HB9 and ISL2) markers, as well as cholinergic markers (CHAT & VACHT).
Gene expression levels were assessed by RT-qPCR, data normalised to HMBS; cDNA samples of the parental human iPSC line (iPSC) were included as reference. Data represents day 11 post-revival samples.
View the step-by-step RNA extraction and RT-qPCR protocol used to generate this data

Co-culture of ioMotor Neurons and ioSkeletal Myocytes
High resolution confocal imaging of ioSkeletal Myocytes (io1002) and ioMotor Neurons wild-type co-culture. Staining with alpha-bungarotoxin (yellow) highlights acetylcholine receptor expression on co-cultured ioSkeletal Myocytes. Desmin (cyan) and microtubule-associated protein 2 (red) define ioSkeletal Myocytes and ioMotor Neurons respectively. Co-culture imaged at day 30, 40X magnification.
Download the step-by-step protocol for culturing ioSkeletal Myocytes and ioMotor Neurons.
Vial limit exceeded
A maximum number of 20 vials applies. If you would like to order more than 20 vials, please contact us at orders@bit.bio.



.png?width=604&name=ioMotor%20Neuron%20Hero%20image%20for%20Webinar%20(1).png)