























cat no | ioEA1006
ioGlutamatergic Neurons TDP‑43 M337V/WT
Human iPSC-derived ALS and FTD disease model
-
Cryopreserved human iPSC-derived cells powered by opti-ox that are ready for experiments in days
-
Excitatory neurons with a disease-relevant mutation in TDP-43 for modelling ALS and FTD
-
Consistent, functional excitatory neurons that form neuronal networks within days

Human iPSC-derived ALS and FTD disease model

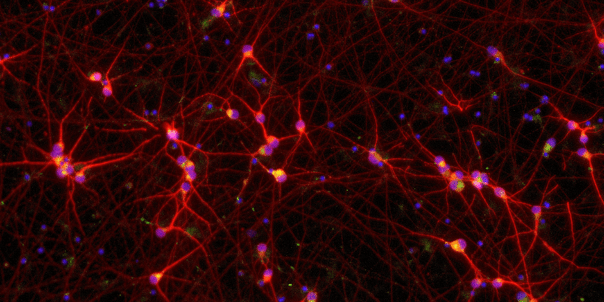
ioGlutamatergic Neurons TDP‑43 M337V/WT express neuron-specific markers comparably to the wild type control
Immunofluorescent staining on day 11 post-revival demonstrates similar homogenous expression of pan-neuronal proteins MAP2 and TUBB3 (upper panel) and glutamatergic neuron-specific transporter VGLUT2 (lower panel) in ioGlutamatergic Neurons TDP‑43 M337V/WT compared to the genetically matched control. 100X magnification.

ioGlutamatergic Neurons TDP‑43 M337V/WT form structural neuronal networks by day 11
ioGlutamatergic Neurons TDP‑43 M337V/WT mature rapidly, show glutamatergic neuron morphology and form structural neuronal networks over 11 days when compared to the genetically matched control. Day 1 to 11 post-thawing; 100X magnification.

ioGlutamatergic Neurons TDP‑43 M337V/WT demonstrate gene expression of neuronal-specific and glutamatergic-specific markers following deterministic programming
Gene expression analysis demonstrates that ioGlutamatergic Neurons TDP‑43 M337V/WT and the wild-type, genetically matched control (WT) lack the expression of pluripotency makers (NANOG and OCT4), at day 11, while robustly expressing pan-neuronal (TUBB3 and SYP) and glutamatergic-specific (VGLUT1 and VGLUT2) markers, and the glutamate receptor GRIA4. Gene expression levels were assessed by RT-qPCR (data normalised to HMBS; cDNA samples of the parental human iPSC line (hiPSC Control) were included as reference). Data represents day 11 post-revival samples, n=2 replicates.
View the step-by-step RNA extraction and RT-qPCR protocol used to generate this data

Disease-related TARDBP is expressed in ioGlutamatergic Neurons TDP‑43 M337V/WT following deterministic programming
Gene expression analysis demonstrates that ioGlutamatergic Neurons TDP‑43 M337V/WT and the wild-type, genetically matched control (WT) express the TARDBP gene encoding TDP‑43. Gene expression levels were assessed by RT-qPCR (data normalised to HMBS; cDNA samples of the parental human iPSC line (hiPSC Control) were included as reference). Data represents day 11 post-revival samples, n=2 replicates.

Differences in neuronal activity between disease models and control cells by MEA analysis indicates the potential of the panel as a relevant translational in vitro drug discovery model for ALS and FTD
Reduced neuronal activity was measured in ioGlutamatergic Neurons TDP-43 M337V/M337V compared to ioGlutamatergic Neurons TDP-43 M337V/WT and the genetically matched control, ioGlutamatergic Neurons.
Microelectrode array (MEA) chips were spotted with 100K (~900K cells/cm2) ioGlutamatergic Neurons (WT), TDP-43 M337V/WT, or TDP-43 M337V/M337V, along with 20K (~180K cells/cm2) human iPSC-derived astrocytes. From DIV6, neurobasal medium was switched to BrainPhys medium by performing a half-medium change every 48 hrs.
Brightfield at 26 DIV (A, left), cells show good coverage of electrodes and produce clear burst and network burst activity as seen in the raster plot of activity (A, right). In the raster plot, each dash indicates a firing event, blue indicates a single electrode burst and the pink box indicates a network burst event.
Quantification of raster plots over the course of culture shows that ioGlutamatergic Neurons TDP-43 M337V/M337V have a lower weighted mean firing rate, and network burst frequency than WT and ioGlutamatergic Neurons TDP-43 M337V/WT (B). No clear difference is noted between WT and TDP-43 M337V/WT. Error bars indicate SEM, n=14 technical repeats.
Data courtesy of Charles River Laboratories.

Identification of splice variants in ioGlutamatergic Neurons and ioMotor Neurons carrying the TDP-43 M337V mutation
To discover neuronal sub-type-specific TDP-43 transcript targets, we used bulk RNA-sequencing to identify alternatively spliced transcripts in ioMotor Neurons and ioGlutamatergic Neurons carrying a heterozygous or homozygous TDP-43 M337V mutation versus wild-type controls. Wild-type and mutant cells were cultured for 30 days in triplicates. RNA samples were collected at day 11, 21 and 30.
Venn diagram comparing the alternatively spliced transcripts at days 21 and 30 that are unique to and shared between ioMotor Neurons and ioGlutamatergic Neurons TDP-43 M337V het and hom genotypes, compared to their respective wild-type controls. Several targets were validated by RT-qPCR analysis, including DPP6, PKIB and PRUNE2. The genes highlighted in bold have previously been associated with FTD/ALS or have been found to be differentially spliced in samples from ALS patients.
View the scientific poster presented at Society of Neuroscience 2025

Schematic representation of the DPP6 splice events occurring in wild-type and TDP-43 mutant ioGlutamatergic Neurons and validation by RT-qPCR
A. UCSC genome browser representation of the human DPP6 locus showing the different splice events (black rectangles) in ioGlutamatergic Neurons. Bulk RNA-seq data showed that splice variant 2 is detected more frequently in day 30 ioGlutamatergic Neurons TDP-43 M337V/M337V mutants while splice variant 1 is more abundant in wild-type control neurons. The curated RefSeq annotations of DPP6 are indicated by the blue lines. The TDP-43 track (red) shows an iCLIP data set (Halleger et al., 2021) intersecting TDP-43 binding sites with the DPP6 transcripts.
B. Graphs showing RT-qPCR analysis of day 30 (D30) ioGlutamatergic Neurons (GN) wild type (WT), TDP-43 M337V/WT (HET) and TDP-43 M337V/M337V (HOM) samples indicated respectively in grey, yellow and orange.
Alternatively spliced genes discovered by bulk RNA-seq in the TDP-43 mutant ioGlutamatergic Neurons and ioMotor Neurons were confirmed by RT-qPCR assays.
View the scientific poster presented at Society of Neuroscience 2025

Phenotypic characterisation of a human iPSC-derived tri-culture using ioGlutamatergic Neurons, ioAstrocytes, and ioMicroglia
Using our fully optimised protocol, ioGlutamatergic Neurons (MAP2, red), ioMicroglia (IBA1, yellow) and ioAstrocytes (vimentin, cyan) were co-cultured to create a highly defined CNS model. High-resolution ICC analysis confirms the successful co-localisation and morphological health of three distinct cell types within a unified environment. By day 7, the protocol yields a highly consistent, integrated network suitable for complex cell modelling. DAPI (blue) highlights the total cell density and integrity of the culture. This protocol is compatible with derivative products of the three cell types, ensuring straightforward implementation across experimental workflows.

Efficient mRNA transfection into ioGlutamatergic Neurons
ioGlutamatergic Neurons are efficiently transfected and show sustained long-term expression of mRNA encoding GFP. ioGlutamatergic Neurons were imaged from day 1 post-thaw and throughout the experiment to assess transfection efficiency and evaluate potential cytotoxic effects of the transfection protocol. Day 1 images were captured prior to transfection on the same day.
Download the step-by-step protocol for lipid-based delivery of synthetic mRNA into ioGlutamatergic Neurons.

Lipid-based delivery of synthetic mRNA into ioGlutamatergic Neurons
ioGlutamatergic Neurons were transfected 24 hours post-thaw using Lipofectamine™ Stem Transfection Reagent. The transfection efficiency was evaluated by fluorescence imaging over 18 days after mRNA delivery, resulting in high transfection efficiency (close to 100%) and long-term sustained GFP expression.
Quantification of the GFP signal shows a decrease in GFP intensity over time, while the percentage of GFP-positive cells remains largely unchanged over time.
(A) The percentage of GFP-positive cells from two independent experiments.
(B) GFP intensity, quantified in successfully transfected cells from two independent experiments is quantified and normalised to day 2 (24 hours post-transfection).

Do more with every vial
The recommended minimum seeding density is 30,000 cells/cm2, compared to up to 250,000 cells/cm2 for other similar products on the market. One small vial can plate a minimum of 0.7 x 24-well plate, 1 x 96-well plate, or 1.5 x 384-well plates. One large vial can plate a minimum of 3.6 x 24-well plates, 5.4 x 96-well plates, or 7.75 x 384-well plates. This means every vial goes further, enabling more experimental conditions and more repeats, resulting in more confidence in the data.
Vial limit exceeded
A maximum number of 20 vials applies. If you would like to order more than 20 vials, please contact us at orders@bit.bio.












_MAP2(R)_Tubb3(B)_Hoechst(B)_20x_merge-comp.jpg?width=604&name=Colour%20webinar%20with%20it-bio%20ioGlutamatergic%20Neurons_VGLUT2(G)_MAP2(R)_Tubb3(B)_Hoechst(B)_20x_merge-comp.jpg)
.png?width=1860&height=1260&name=bit.bio_3x2_ioGlutamatergic%20Neurons_MAP2_Hoescht_x20_hi.res%20(1).png)






