20.02.2023 | Published by bit.bio
.png)
Summary
This blog shows you how to prevent detachment, improve neuronal adherence, and achieve reproducible glutamatergic neuronal cultures using 4 of our top tips.
By combining careful handling, precise media changes, and optimised cell density with a defined Geltrex Flex + poly-D-lysine coating protocol, researchers can ensure cell culture neurons adhere evenly, remain viable, and develop stable networks suitable for imaging, molecular, or electrophysiological analysis. Reproducible culture results start with good habits; with consistent technique and routine care, experimental reproducibility and data quality can be greatly improved.
Human iPSC-derived glutamatergic neurons: Cell culture hacks and a Geltrex Flex + poly-D-lysine coating protocol
Human iPSC-derived excitatory neurons provide helpful physiologically relevant cell models to investigate neurodevelopmental and neurodegenerative disorders, although achieving healthy neuronal cultures can be challenging. bit.bio central nervous system (CNS) scientist Dr Kaiser Karim has studied the genomic mechanisms governing reprogramming of human iPSCs into glutamatergic neurons and contributed to research investigating the effects of energetic substrate availability on global neural network behaviour.
In this blog, Kaiser shares his top tips for overcoming common challenges with glutamatergic cell culture neurons, including how to apply a Geltrex Flex + poly-D-lysine coating protocol to improve adhesion and promote long-term neuronal network stability. By using careful handling to prevent cell detachment, perfecting routine media changes, and using the right cell density, it’s possible to achieve regular and consistent cell culture success!
Go straight to the top tips!
- Top tip #1: Careful handling can prevent cell detachment from culture surfaces
- Top tip #2: Geltrex Flex + poly-D-lysine coating protocol to enhance cell adherence
- Top tip #3: Closely monitor media change timings to ensure successful cultures
- Top tip #4: Use the right cell density to get better-quality data from your experiments
What are glutamatergic neurons?
Glutamatergic neurons are cells that produce the common excitatory neurotransmitter glutamate, ‘exciting’ other neurons. These cells can be found throughout the CNS, for example within the cortex, where they are abundant, as well as the spinal cord and hypothalamus. Glutamatergic neurons contribute to a number of CNS-related functions such as cognition, memory, learning, and sensory perception1. They have also been implicated in a number of neurodegenerative diseases, including Alzheimer’s disease2.
The human iPSC-derived ioGlutamatergic Neurons provided by bit.bio are reprogrammed iPSC-derived cells and provide a highly relevant and consistent in vitro human model3. They form networks of postsynaptic dendrites and presynaptic axon projections, recapitulating the type of human neuron-to-neuron communication networks found in the body4.
"With such complex and fragile structures, culturing glutamatergic neurons in vitro requires advanced planning and special attention", cautions Kaiser.
Following a consistent Geltrex Flex + poly-D-lysine coating protocol for better cell adherence is just one way to improve reproducibility. Here are our top tips for success.
Top tip #1: Careful handling can prevent cell detachment from culture surfaces
Cell detachment is a common problem scientists face when culturing glutamatergic neurons. ioGlutamatergic Neurons, for example, can form neurites after only two days in culture and are ready for experiments in as little as 11 days, although they can be fragile. “As neuronal cell bodies form connections across the culture plate via neurites, the disruption of one neuron can impact the entire culture,” says Kaiser. With careful handling, however, the development of these neuronal networks will increase and mature over time.
There are 4 key considerations surrounding medium changes that can reduce the risk of cell detachment:
-
Avoid aspirating too close to neurons, as this can potentially create a ‘tear’ in neuronal cultures. Kaiser comments: “Unfortunately, once there is a tear and disruption to the culture, the glutamatergic neurons will suffer permanent damage and eventually peel off.” The early stages of glutamatergic neuron culture can be especially vulnerable, as 90% medium changes are required. Cells can be disrupted if the media is changed too quickly or aspiration occurs too close to the neurons.
-
Every time you add medium to your glutamatergic neuron cultures, target a different location within the plate or well, and keep track of where it’s been added. For example, on Day 0, you could add medium to the left side of the culture well, Day 2 on the right side, Day 4 at the top, etc. The cells’ neuronal networks are more likely to detach if they are continuously fed in the same place. This is because the pressure of the liquid going into the plates/wells can weaken cell attachment as well as cells’ extracellular matrix (ECM).
-
Another consideration is tilting the plate to aspirate spent medium from the culture wells when carrying out a 90% medium change. This helps to ensure that the pipette tip doesn’t touch the culture.
-
Finally, if using a stripette, set it at a very low flow rate to prevent disruption of the neuronal network, gently handling both the cell culture plate and liquid flow with careful attention at all times.
Careful handling is essential for preventing detachment; however, the long-term stability of neuronal cultures also depends on proper surface preparation. Using a defined Geltrex Flex + poly-D-lysine protocol helps establish strong, uniform cell adhesion. Here’s how to optimise it.
 Careful handling and media changes that avoid causing a ‘tear’ in the culture can prevent cells from lifting off surfaces and improve glutamatergic neuronal cell culture success. Tilting the cell culture plate, as shown here, can help prevent direct damage to the cells caused by adding fresh medium to wells.
Careful handling and media changes that avoid causing a ‘tear’ in the culture can prevent cells from lifting off surfaces and improve glutamatergic neuronal cell culture success. Tilting the cell culture plate, as shown here, can help prevent direct damage to the cells caused by adding fresh medium to wells.
Top tip #2: Optimise your Geltrex Flex + PDL coating protocol to enhance cell adherence
Human iPSC-derived neuronal cultures depend on robust cell attachment to thrive. Coating the culture surface with poly-D-lysine before adding extracellular matrix (ECM) components helps create an environment that supports healthy, functional cell culture neurons.
After much testing and optimisation, we’ve found that combining a defined poly-d-lysine coating protocol with Geltrex Flex works very successfully for iPSC-derived human neurons. Poly-D-lysine (PDL) is a synthetic, non-ECM coating molecule that improves cell attachment to surfaces via electrostatic interactions. To support ioGlutamatergic Neurons, we resuspend PDL in borate buffer rather than water — a simple yet critical adjustment to the standard PDL protocol that significantly improves coating efficiency and stability. Geltrex is a soluble basement ECM-derived coating that contains a cocktail of laminin, collagen IV, heparin sulphate proteoglycans, and entactin/nidogen. The presence of such molecules plays a role in recapitulating the physiological microenvironment, promoting cell-cell interactions and network activity5,6. With this double-layer surface coating, ioGlutamatergic Neurons can be cultured for over 2-3 weeks, to enable functional studies into neuronal maturation and activity, explains Kaiser.
Geltrex Flex + poly-D-lysine coating protocol for glutamatergic neuronal cultures
Purpose: This Geltrex Flex + poly-D-lysine coating protocol provides a simple method for preparing charged surfaces that promote the adhesion and survival of human iPSC-derived neurons. Using an optimised Geltrex Flex + poly-D-lysine coating protocol helps ensure even coating across wells or coverslips, improving culture uniformity and experimental reproducibility.
PDL coating solution preparation
-
Make up 50 mL of 1X borate buffer by diluting 2.5 mL of the 20X stock with 47.5 mL of sterile water.
-
Resuspend a 5 mg vial of PDL in 50 mL of the 1X borate buffer, for a working concentration of 100 μg/mL.
-
PDL coating solution can be kept at -20°C for long-term storage.
-
Tip: To avoid freeze-thaw cycles, aliquot the coating solution as appropriate for future use.
-
PDL coating
-
Calculate the total surface area to be coated. We recommend the coating volumes shown in the table below (circa 100 μL per cm2).
|
Coating solution |
384 well |
96 well |
24 well |
12 well |
6 well |
|
PDL |
15 μL |
50 μL |
400 μL |
500 μL |
1 mL |
- Coat the surface of your cell culture vessel with the PDL coating solution.
- Incubate the coated plates overnight at 37°C or for at least 3 hours.
- Aspirate PDL solution and then wash 3 times with sterile water.
- Tip: For each wash, use the same volume used for coating.
- Aspirate the water and allow coated surfaces to dry completely in a laminar flow hood (without lids). This typically requires 30 to 60 minutes.
Geltrex Flex preparation
-
Remove Geltrex Flex stock from -80°C and thaw on ice in a 4°C fridge overnight.
-
The next day, prepare single-use aliquots according to foreseen use. Store at -80°C to minimise further freeze-thawing.
-
Depending on the volume, the smaller aliquots should take about 30 minutes to thaw while kept on ice.
-
Tip: Geltrex Flex polymerises quickly at temperatures above 4°C - always keep Geltrex Flex below 4°C.
-
Geltrex Flex coating
-
Calculate the total surface area to be coated.
-
Dilute the Geltrex Flex at 0.15 mg/mL in chilled DMEM/F-12.
-
Coat the surface area of your culture vessel with the Geltrex Flex:DMEM/F-12 coating solution. We recommend the following coating volumes (circa 100 μL per cm2):
|
Coating solution |
384 well |
96 well |
24 well |
12 well |
6 well |
|
Geltrex Flex: DMEM/F-12 |
15 μL |
50 μL |
400 μL |
500 μL |
1 mL |
-
Incubate the coated plates at 37°C for a minimum of 60 minutes.
-
Carefully aspirate off the excess Geltrex Flex, then immediately plate the cells.
Top tip #3: Closely monitor media change timings to ensure successful cultures
Once plates have been successfully coated with poly-D-lysine-Geltrex Flex and neurons are securely attached, consistent media changes become essential to sustain healthy neuronal cultures. Even with an optimal surface coating, irregular feeding schedules or abrupt changes in medium composition can stress cells and compromise network formation. For ioGlutamatergic Neurons, Dr Kaiser Karim recommends a defined workflow that balances nutrient renewal with minimal physical disruption. After thawing the cells and seeding them onto coated plates, allow a four-day stabilisation phase in the presence of doxycycline (dox). During this time, 90% medium changes are necessary every 48 hours, taking care to aspirate gently and avoid disturbing the developing networks.
For example:
-
Day 0 (Monday): Thaw and seed neurons onto PDL + Geltrex Flex-coated plates.
-
Day 2 (Wednesday): Perform the first 90% media change (include dox and DAPT).
-
Day 4 (Friday): Final 90% media change (omit dox).
After stabilisation, begin the maturation phase by replacing 50% of the medium every 48 hours from Day 6/7 onwards. This gradual reduction in media replacement supports metabolic equilibrium and reduces mechanical stress.
“This protocol is also helpful for those times when ‘life happens’, and you can’t make it into the lab on the weekend to do a media change,” Kaiser adds. Maintaining a flexible yet consistent schedule helps preserve the morphology and viability of developing networks, ensuring that your cell culture neurons remain healthy and reproducible across experiments.
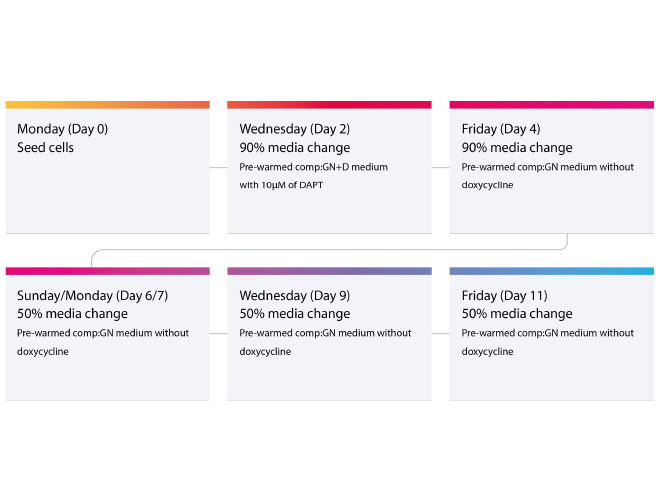 Suggested media change cycle for bit.bio’s iPSC-derived glutamatergic neurons. This workflow can be repeated for the length of cell culture time. Further details can also be found in the bit.bio ioGlutamatergic Neurons’ User Manual.
Suggested media change cycle for bit.bio’s iPSC-derived glutamatergic neurons. This workflow can be repeated for the length of cell culture time. Further details can also be found in the bit.bio ioGlutamatergic Neurons’ User Manual.
Top tip #4: Use the right cell density to get better-quality data from your experiments
Seeding density is one of the most influential factors in the success of neuronal cultures. Even with regular media changes and an optimised Geltrex Flex + poly-D-lysine coating protocol, overcrowding or under-seeding can affect cell survival, network formation, and data reproducibility.
According to Dr Kaiser Karim, using the right density for your assay type ensures that neurons develop evenly, making dendrites, axons, and synaptic connections easier to image, analyse, and maintain. High densities (>100,000 cells/cm²) often make it difficult to isolate individual neurons for downstream assays such as immunocytochemistry (ICC) or RNAseq.
bit.bio’s ioGlutamatergic Neurons can be seeded out at a much lower density — as few as 30,000 cells/cm2 — while maintaining excellent viability and functional connectivity.
At this density, ioGlutamatergic Neurons can be used in routine experiments and have been pre-validated for immunocytochemistry, qPCR, and RNAseq. The cells have also been validated for morphology characterisation, including brightfield imaging and quantitative live-cell imaging and analysis (like neurite outgrowth). Furthermore, at this density, the cells are cost-effective without impacting quality.
“For complex experiments, such as analysing neuronal network activity using neuronal electrophysiology or calcium signalling, higher seeding densities may be necessary, including additional optimisation,” adds Kaiser.
For more tips and information on ioGlutamatergic Neurons cell culture, check out this helpful step-by-step video with Kaiser.
Do you have any cell culture tips to share? We’d love to hear from you!
About Kaiser Karim, PhD
Kaiser completed his PhD in Clinical Neurosciences at the University of Cambridge, under the supervision of Dr Mark Kotter (NIHR Clinician Scientist, Consultant in Neurosurgery, University of Cambridge and CEO bit.bio), where he studied the genomic mechanisms that govern reprogramming of human iPSCs into induced glutamatergic neurons. This work followed the pivotal 2017 paper published by Mark Kotter’s academic research group. He has also contributed to work looking into the effects of energetic substrate availability on global neural network behaviour. Kaiser’s research has provided a deeper understanding of bit.bio’s ioGlutamatergic Neurons and helped lay the foundations for its commercialisation.
 Kaiser KarimScientist at bit.bio
Kaiser KarimScientist at bit.bio
References
- Sanacora G, Zarate C, Krystal J et al. Targeting the glutamatergic system to develop novel, improved therapeutics for mood disorders. Nat Rev Drug Discov 7, 426–437 (2008). https://doi.org/10.1038/nrd2462.
- Gasiorowska A, Wydrych M, Drapich P, et al. The Biology and Pathobiology of Glutamatergic, Cholinergic, and Dopaminergic Signaling in the Aging Brain. Front Aging Neurosci. 2021 Jul 13;13:654931. doi: https://doi.org/10.3389/fnagi.2021.654931.
- Pawlowski M, Ortmann D, Bertero A, et al. Inducible and Deterministic Forward Programming of Human Pluripotent Stem Cells into Neurons, Skeletal Myocytes, and Oligodendrocytes. Stem Cell Reports. 2017 8(4):803-812. https://doi.org/10.1016/j.stemcr.2017.02.016.
- Tourigny DS, Abdul Karim MK, Echeveste R, & Kotter, RN. Energetic substrate availability regulates synchronous activity in an excitatory neural network. PLOS ONE. 2019 14(8), e0220937. https://doi.org/10.1371/journal.pone.0220937.
- Barros CS, Franco SJ, Müller U. Extracellular matrix: functions in the nervous system. Cold Spring Harb Perspect Biol. 2011 1;3(1):a005108. https://doi.org/10.1101/cshperspect.a005108.
- Lam D, Enright HA, Cadena J et al. Tissue-specific extracellular matrix accelerates the formation of neural networks and communities in a neuron-glia co-culture on a multi-electrode array. Sci Rep. 2019 9: 4159. https://www.nature.com/articles/s41598-019-40128-1.