12.07.2023 | Published by bit.bio
- Models will be used to investigate, screen for and develop therapies for the restoration of dystrophin, the protein that is missing in DMD patients
- Researchers can also use the models to better understand DMD and how the disease affects the mechanisms of skeletal muscle cells
- The new bit.bio DMD disease models offer consistency, scalability and reproducibility, overcoming the challenges associated with the use of prior models, such as primary muscle cells and immortalised cell lines
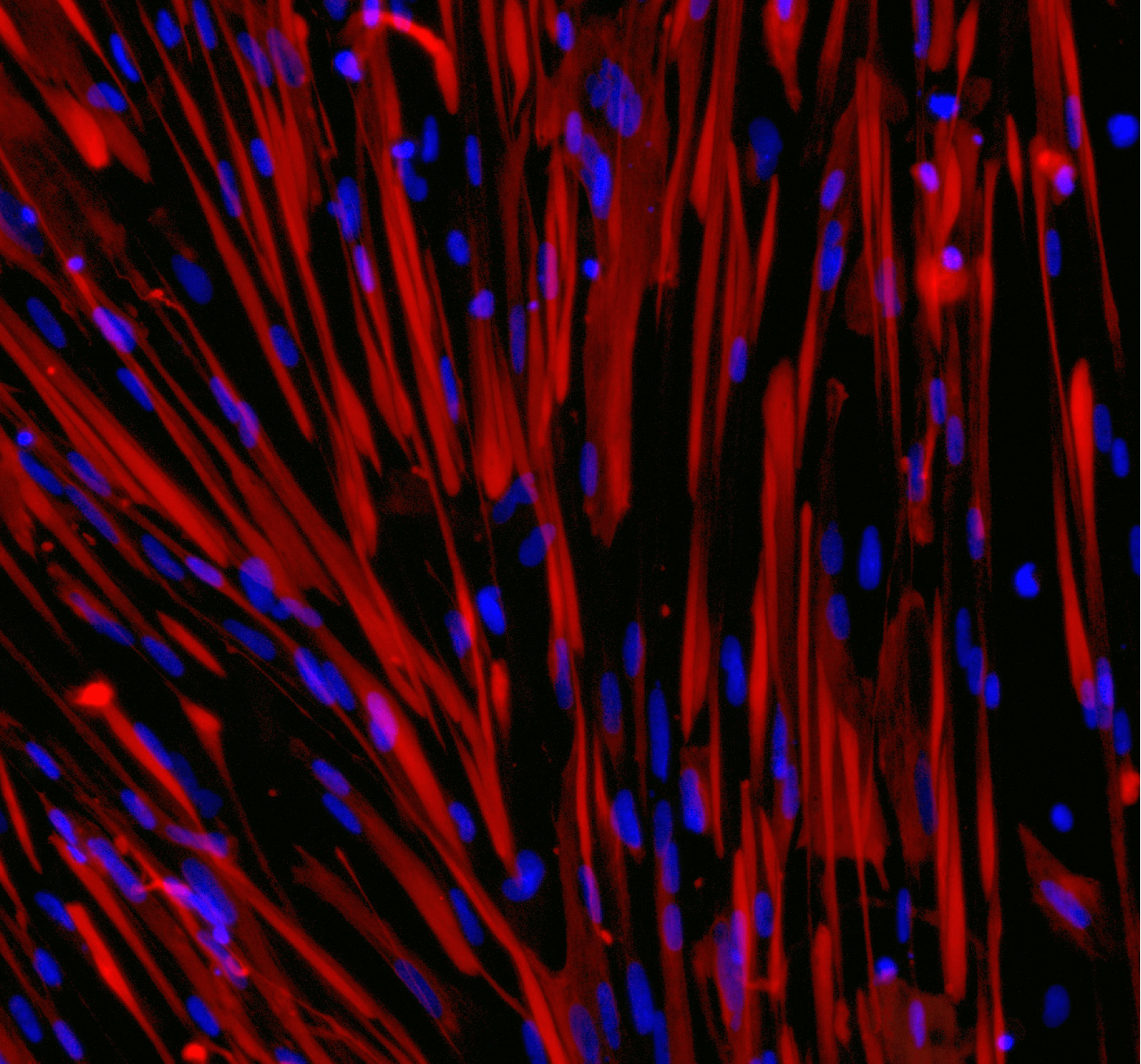
CAMBRIDGE, UK 12:00, 12 July 2023: bit.bio, the company coding human cells for novel cures, today launches its first set of muscle cell disease model products designed to advance the discovery and development of treatments for Duchenne muscular dystrophy (DMD). The disease models, ioSkeletal Myocytes DMD Exon 44 Deletion™️ and ioSkeletal Myocytes DMD Exon 52 Deletion™️, are human skeletal myocytes that carry a genetically engineered deletion of exon 44 or exon 52 in the gene that encodes for the dystrophin protein. Cells have been precision reprogrammed from induced pluripotent stem cells (iPSCs) with bit.bio’s opti-ox™️ technology.
DMD affects 1 in 3,500 male births worldwide. The average life expectancy is 27 years with muscle weakness appearing in early childhood and worsening rapidly. It is caused by genetic mutations or deletions that prevent the production of functional dystrophin - part of a group of proteins that work together to strengthen muscle fibres and protect them from injury as muscles contract and relax. While over a thousand different mutations in the dystrophin gene can cause the disease, the deletions in the two ioSkeletal Myocytes DMD disease models launched by bit.bio today are amongst the most common.
bit.bio’s ioSkeletal Myocytes DMD disease models support the development of new treatments for DMD patients. At present, human or mouse primary cells or immortalised cell lines are used. However, human primary cells show inherent variability due to donor-to-donor differences and are difficult to source; immortalised cell lines often have genetic alterations that affect their behaviour, and mouse tissue may not represent human biology. bit.bio’s ioSkeletal Myocytes DMD disease models address these challenges by providing a consistent and scalable source of human cells.
DMD is a complex and relentless condition and a major challenge. Despite recent advances, I know from personal experience that many unmet clinical needs remain. bit.bio’s mission is to code cells for novel cures. My sincere hope is that this latest launch in our ioCell research product range will support and accelerate the development of effective treatments for DMD. Today’s launch is a further proof point of bit.bio’s unique and rapidly accelerating ability to add new products to our portfolio."
 Mark KotterCo-founder and CEO, bit.bio
Mark KotterCo-founder and CEO, bit.bio
ioSkeletal Myocytes DMD disease models were developed as part of bit.bio’s partnership with Charles River Laboratories International, Inc., a highly respected, global provider of drug discovery and non-clinical development solutions. Charles River has evaluated the bit.bio DMD disease models for drug discovery applications, including studies showing that the DMD disease cells lack expression of dystrophin, making them an ideal model for exon skipping applications.
In addition, Charles River also showed that dystrophin expression was restored in cells with deletion of exon 44 when treated with an antisense oligonucleotide (ASO) therapy. ASO therapies use single-stranded oligonucleotides that can alter RNA expression and splicing and thereby reduce, restore, or modify protein expression. In the case of DMD, several ASO therapies are in development that aim to restore a truncated but functional dystrophin protein to improve muscle function in patients.
"ioSkeletal Myocytes DMD disease models are an important step towards the development of new treatments for DMD. Together with the state-of-the-art drug discovery technologies and expertise in our teams we are now able to help our clients to demonstrate mechanistic, and in the future hopefully also functional effects of potential new drugs, at an early stage of drug development."
 Marijn Vlaming PhDCharles River Head of Discovery Sciences
Marijn Vlaming PhDCharles River Head of Discovery Sciences
ioSkeletal Myocytes DMD disease models are the eighth and ninth products launched by bit.bio this year under its ioCells™ brand - human cells and disease models for research and drug discovery. Within bit.bio’s existing portfolio, wild type ioSkeletal Myocytes™ provide a genetically matched control to the DMD disease models. As ioDisease Model™ cells come from an identical human genetic background to ioWild Type™ cells, any experimental differences observed between the wild type and the disease model can be confidently attributed to the effects caused by the disease-specific mutations.
"These new disease models provide scientists with a consistent and scalable source of cells to study how exon deletions impact muscle cell function, and enable investigation of therapeutic candidates for dystrophin restoration. Their suitability for use in multi-well plate formats means screening experiments can be conducted in a physiologically relevant human cell model on a larger scale, overcoming some of the limitations associated with the use of animal models and primary cells. Having access to a relevant translational model for DMD will help accelerate the work of researchers developing treatments for this relentlessly progressive degenerative disease."
 Farah Patell-SochaVP Research Products
Farah Patell-SochaVP Research Products
The products are now available to order on the bit.bio website:
ioSkeletal Myocytes DMD Exon 44 Deletion
ioSkeletal Myocytes DMD Exon 52 Deletion