29.09.2022 | Published by bit.bio
29.09.2022 | Published by bit.bio

bit.bio adds two new human cell products to address the translation gap and accelerate research and drug discovery for neurodegenerative disease
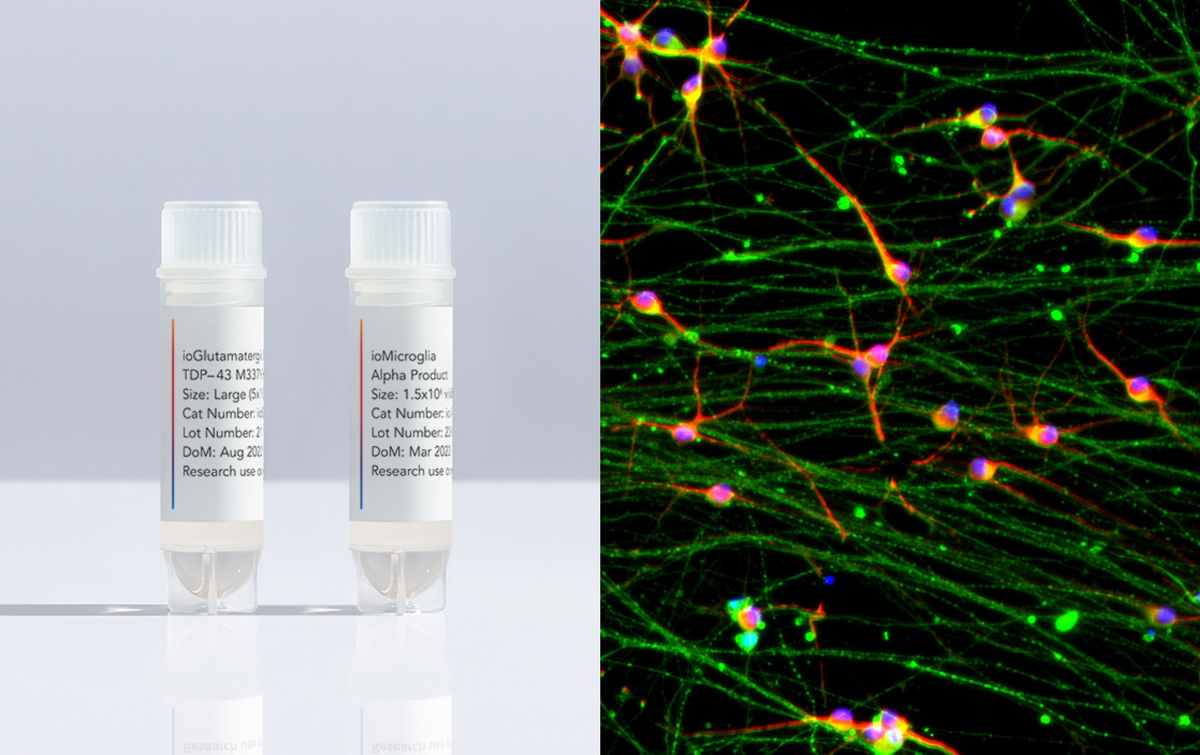
CAMBRIDGE, 29th September 2022 — Cell coding company bit.bio has announced an expansion to its product portfolio - ioGlutamatergic Neurons TDP-43 M337V disease model and early access to its ioMicroglia cell product.
Despite considerable research efforts and funding, the development of therapies for devastating diseases like Alzheimer’s disease (AD), Frontotemporal dementia (FTD) and amyotrophic lateral sclerosis (ALS) has been challenging. Due to a lack of standardised, easy to use and readily accessible human cell models, scientists have relied on animal models and cell lines that differ considerably from human biology.
bit.bio's latest cell products provide a scalable source of human cells and will enable scientists to study neurodegenerative diseases in a human context. With consistency across batches and a scalable supply, bit.bio’s products will significantly reduce experimental variability in non-clinical studies and improve the translatability and reproducibility of research findings. These unique product features have the potential to transform research and drug discovery.
“The products we are announcing today address an area of high unmet clinical need where high failure rates in drug development are common and no effective treatments exist. I look forward to seeing how our customers will use them to develop new insights and treatments for these devastating conditions.
"This is another step towards our vision of an exciting future in which precision reprogrammed human cells will accelerate biomedical innovation and a new generation of cures. The launch of two new cell products for research and drug discovery in neurodegenerative diseases validates our cell identity coding platform’s ability to create and manufacture any human cell type consistently at scale." Mark KotterCo-founder and CEO, bit.bio
Mark KotterCo-founder and CEO, bit.bio
Today, AD and FTD, the leading causes of early onset dementia, have no treatment options to stop or slow their onset. Similarly, current treatment options for ALS, the most common motor neuron degenerative disease, are limited. ioGlutamatergic Neurons TDP-43M337V, have a mutation in the TAR DNA binding protein gene that codes for the TDP-43 protein, which is known to cause both FTD and ALS. The disease model cells and the genetically matched control, ioGlutamatergic Neurons, mature rapidly, are highly reproducible between batches, and have unprecedented scalability. These key features make them ideally suited to high-throughput screening applications for early drug discovery. Being able to compare data from the physiologically-relevant disease models to those of the control offers the potential to identify and investigate the effects of the genetic mutation on the disease mechanisms of FTD and ALS.
Increasing evidence suggests that microglia contribute to the onset and progression of AD and are involved in the pathogenesis of ALS and FTD. Therefore they may represent an additional therapeutic target. However, speed, variability, and scalability continue to be major challenges with commonly used microglia. ioMicroglia, now available as part of an early access program, address these challenges allowing scientists to work with consistent, functional cells that are ready for experimentation within just 10 days.
“Human cells are key to disease research, drug discovery, and clinical translation. However, traditional methods of producing human cells have long, laborious, protocols that often result in heterogenous cell populations and can lead to data variability. Our latest products provide robust, standardised tools for neurodegenerative research and drug discovery, paving the way for high-throughput screening and drug target validation in human iPSC-derived models that was previously impossible, and bringing huge benefits to medicine as a result.”
 Farah Patell-Sochabit.bio VP Research Products
Farah Patell-Sochabit.bio VP Research Products
The latest disease models are now available to order.
Register your interest and find full physiological data for early access ioMicroglia vials.