





























cat no | io1002
ioSkeletal Myocytes
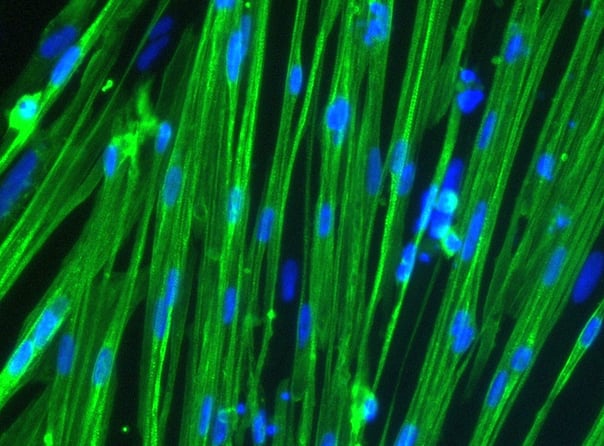
Human iPSC-derived skeletal myocytes
-
Cryopreserved human iPSC-derived cells powered by opti-ox that are ready for experiments in days
-
Ideal for the study of skeletal muscle and modelling DMD and other neuromuscular disorders
-
Functional cells contract in response to chemical and electrical stimuli in 2D and 3D microtissues

Human iPSC-derived skeletal myocytes
ioSkeletal Myocytes generated by transcription factor-driven deterministic cell programming of iPSCs using opti-ox technology
Time-lapse video capturing the rapid and homogeneous skeletal myocytes phenotype acquisition upon thawing of cryopreserved ioSkeletal Myocytes. 10 day time course.

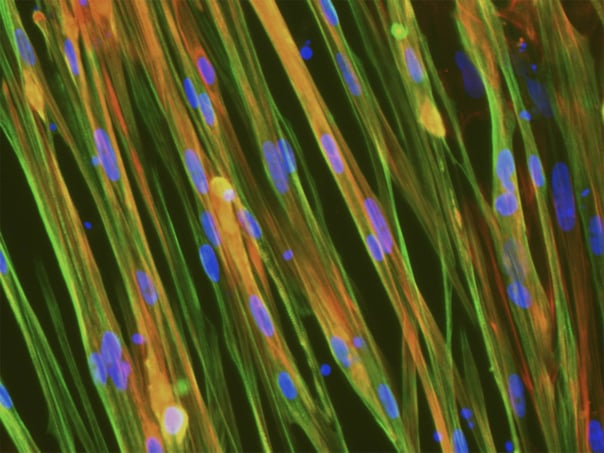
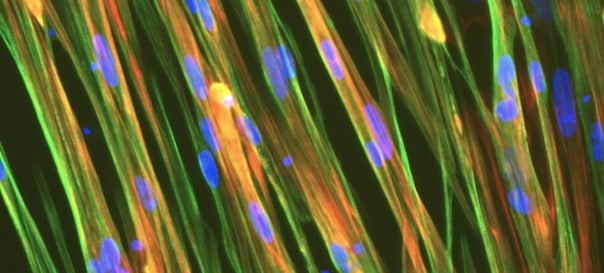
Cells demonstrate classical myocyte morphology
ioSkeletal Myocytes form elongated multinucleated myocytes over 10 days. Day 3 to 10 post-thaw; 10X magnification.
Video showing spontaneous, synchronised calcium waves
ioSkeletal Myocytes were seeded at 100,000 cells/cm2 in black 24-well ibiTreat plates and cultured according to the user manual. On day 10 post-thaw, cells were loaded with Fluo4-AM for the assessment of spontaneous calcium transients.
The video shows spontaneous, synchronised calcium waves; signal intensity increases in proportion to intracellular Ca2+ concentration.

Spontaneous synchronised calcium waves
ioSkeletal Myocytes loaded with Fluo4-AM for the assessment of spontaneous calcium transients.
Left: still images of one calcium wave captured across the culture;
Right: Graph showing spontaneous, synchronised calcium waves. Each coloured line represents Fluo4-AM intensity over time in one of nine circular regions of interest distributed evenly across the field of view, illustrating synchronicity of calcium transients across different parts of the field of the culture.

Contraction in response to increased extracellular potassium levels
(A) Immunofluorescence staining of ioSkeletal Myocytes revealing robust expression of sarcomere structures.
Contraction is stimulated by depolarisation of the cells using potassium chloride (KCl), and the consequent increase in intracellular calcium (Ca2+) is detected using calcium binding indicator dye Indo-1 AM.
(B) Representative images of ioSkeletal Myocytes incubated with Indo-1 AM (5 µM) and 0.02% Pluronic F127; cells were excited at UV spectra (355 nm).
(C) Changes in Indo-1 AM ratio shows Ca2+ influx induced by 45 mM KCl.
Data courtesy of Gabriel E. Valdebenito and Michael R. Duchen, 2021. UCL, UK

Contraction in response to electrical stimulation
Contraction is induced by electrical stimulation and the cells release and sequester Ca2+.
The ioSkeletal Myocytes can withstand repeated electrical stimulation while maintaining their ability to regulate intracellular calcium signalling. Electrical stimulation, 2 Hz, 6 v, 2 ms.
Data courtesy of Gabriel E. Valdebenito and Michael R. Duchen, 2021. UCL, UK

Muscle bundles express muscle cell markers and show increasing maturity over time
(A) SEM image of ioSkeletal Myocytes muscle microtissues on day 14 cultured in 3D on a MUSbit microchip (Bi/ond), which includes pillars designed for anchoring muscle cell bundles; green arrow indicates muscle fibers.
(B) The cells were cultured over 14 days and expressed muscle cell markers, sarcomeric alpha actinin (SAA) and actin; a higher degree of cross-striation of SAA is seen on day 14 (yellow arrows).
Data courtesy of M. Han and M. Aarts, formerly at Bi/ond Solutions BV.

Functional 3D muscle bundles respond to electrical stimuli
(A) Twitch (black) and tetanic (red) forces are observed at day 7 for one bundle (left) and several bundles (right).
(B) Contractile force increases in one bundle (left) and several bundles (right) from day 7 to day 14, indicating muscle bundles become stronger and more mature over time.
Data courtesy of M. Han and M. Aarts, formerly at Bi/ond Solutions BV.

Functional 3D muscle bundles respond to pharmacological stimuli
(A) Contraction is inhibited when the 3D muscle cell bundle is electrically stimulated following treatment with BDM, a non-selective skeletal muscle myosin-II ATPase inhibitor.
(B) Contractility is increased when the bundle is electrically stimulated following addition of caffeine, which stimulates Ca2+ release from the sarcoplasmic reticulum.
Data courtesy of M. Han and M. Aarts, formerly at Bi/ond Solutions BV.

Statin-induced myopathy is recapitulated in 3D muscle cell bundles
ioSkeletal Myocytes were treated with a range of cerivastatin concentrations.
(A) Cerivastatin causes damage to the muscle bundle that results in reduced contraction amplitude. Bundles cultured on the MUSbit chip (Bi/ond) were treated with increasing doses of cerivastatin from day 7-14. Tetanic stimulation of 20 Hz for 1s. n=6 for DMSO, n=4 per cerivastatin concentration. Dunnett's one-way ANOVA statistical analysis: ** indicates P < 0.01, **** indicates P<0.0001.
(B) Representative images of muscle cell bundles at day 12. Cerivastatin treatment resulted in loss of myofiber organisation.
Data courtesy of M. Han and M. Aarts, formerly at Bi/ond Solutions BV.

Cells demonstrate gene expression of key myogenic markers following deterministic cell programming
Following deterministic cell programming, ioSkeletal Myocytes downregulate expression of the pluripotency genes (A), while demonstrating robust expression of key myogenic markers (B). Gene expression levels were assessed by RT-qPCR (data normalised to HMBS; cDNA samples of the parental human iPSC line (hiPSC) were included as reference). Data represents day 10 post-revival samples; n=7 biological replicates.
View the step-by-step RNA extraction and RT-qPCR protocol used to generate this data

Whole transcriptome analysis demonstrates high lot-to-lot consistency across three manufactured lots of ioSkeletal Myocytes
Bulk RNA-sequencing analysis was performed on three different lots of ioSkeletal Myocytes on day 1 and day 10 post-revival. A principal component analysis (PCA) to assess gene expression variance between three different manufactured lots showed tight clustering of the samples at the day 10 time point, demonstrating high consistency between these lots. This lot-to-lot consistency of ioSkeletal Myocytes will help reduce experimental variation and increase the reproducibility of experiments.
Colours represent the three lots of products and the parental non-induced hiPSC line; shapes represent the different culture timepoints.

Co-culture of ioSkeletal Myocytes and ioMotor Neurons
High resolution confocal imaging of ioSkeletal Myocytes and ioMotor Neurons (io1027) co-culture. Staining with alpha-bungarotoxin (yellow) highlights acetylcholine receptor expression on co-cultured ioSkeletal Myocytes. Desmin (cyan) and microtubule-associated protein 2 (red) define ioSkeletal Myocytes and ioMotor Neurons respectively. Co-culture imaged at day 30, 40X magnification.
Download the step-by-step protocol for culturing ioSkeletal Myocytes and ioMotor Neurons.

Efficient mRNA transfection into ioSkeletal Myocytes
ioSkeletal Myocytes are efficiently transfected and show sustained long-term expression of mRNA encoding GFP. ioSkeletal Myocytes were imaged from day 1 post-thaw and throughout the experiment to assess transfection efficiency and evaluate potential cytotoxic effects of the transfection protocol. Day 3 images were captured prior to transfection on the same day.
Download the step-by-step protocol for lipid-based delivery of synthetic mRNA into ioSkeletal Myocytes.
Vial limit exceeded
A maximum number of 20 vials applies. If you would like to order more than 20 vials, please contact us at orders@bit.bio.













.png?width=604&name=ISSCR24-DMD-Exon44-3D-muscle-bundle%20(1).png)





.jpeg?width=604&name=New%20Project%20(13).jpeg)








/ioSkeletal-Myocytes-DMD-Exon-52-Deletion-hero.webp?width=1276&height=1190&name=ioSkeletal-Myocytes-DMD-Exon-52-Deletion-hero.webp)

